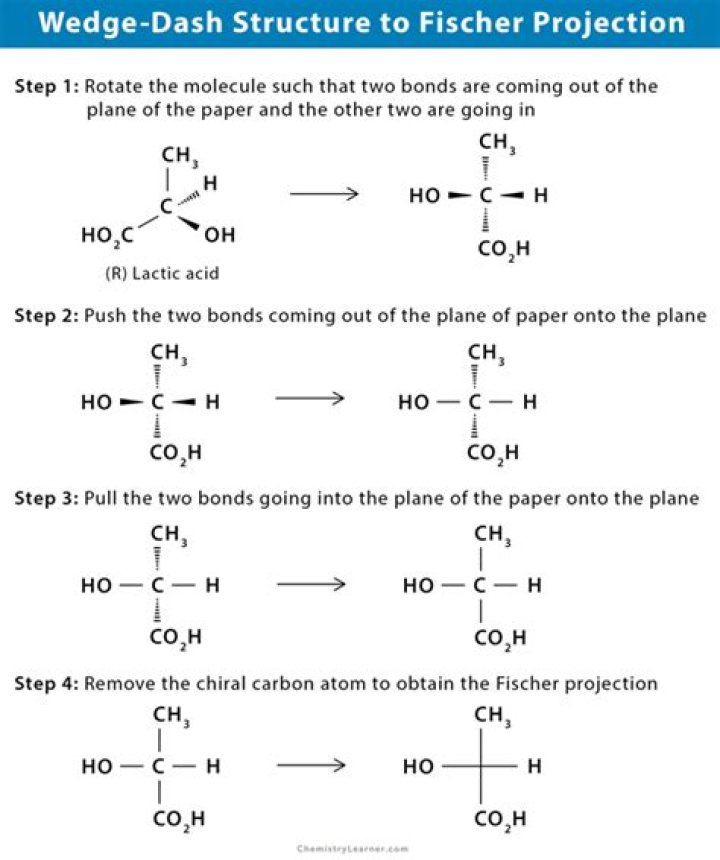
A formal two-dimensional representation of a three-dimensional molecular structure obtained by projection of bonds (symbolized as lines) onto a plane with or without the designation of the positions of relevant atoms by their chemical symbols..
Also to know is, what is projection formula?
Definition of projection formula. : a perspective formula projected so as to represent it in two dimensions — compare structural formula.
Also, what is Fischer projection in chemistry? The Fischer projection, devised by Emil Fischer in 1891, is a two-dimensional representation of a three-dimensional organic molecule by projection. Fischer projections were originally proposed for the depiction of carbohydrates and used by chemists, particularly in organic chemistry and biochemistry.
Herein, what is sawhorse projection in chemistry?
A sawhorse projection is a view of a molecule down a particular carbon-carbon bond.
What is Newman projection formula?
Newman. projection. 3D structure. A Newman projection, useful in alkane stereochemistry, visualizes the conformation of a chemical bond from front to back, with the front atom represented by a dot and the back carbon as a circle. The front carbon atom is called proximal, while the back atom is called distal.
Related Question Answers
What is the formula of unit vector?
Where, v is magnitude of
vector. Example: Find the direction of the
vector v= <9, 12=>? Use the
formula to find the magnitude of v.
Solution:
| FORMULAS Related Links |
| Continuous Compound Interest Formula | Celsius To Farenheit Formula |
What is vector projection used for?
Vector projections are used for determining the component of a vector along a direction. Let us take an example of work done by a force F in displacing a body through a displacement d. It definitely makes a difference, if F is along d or perpendicular to d (in the latter case, the work done by F is zero).What is gauche Newman projection?
If there is a chain of carbon as an attachment in both the front and back carbon, and the attachments are 60 degrees apart, this is considered a gauche conformation. If the carbon attachments are on opposite ends, 180 degrees apart, this is considered an anti conformation.Which best describes a sawhorse projection?
Which BEST describes a sawhorse projection? This projection views the carbon-carbon bond at an angle and, by showing all the bonds and atoms, their spatial arrangements are indicated.What is conformation in chemistry?
Definition of Conformation. What is Conformation? The spatial arrangement of the atoms affording distinction between stereoisomers which can be interconverted by rotations about formally single bonds.What is conformational analysis in organic chemistry?
Conformational analysis deals with the shapes that molecules can adopt by rotations about single bonds. These structures are called conformational isomers or conformers. Conformational isomers are interconvertable isomers of the same molecule that result from rotations of single bonds.Which Newman projection is most stable?
The most stable one would have the rear hydrogen in between the front methyl and bromine in a staggered conformation to minimize [lone-pair]-[bonding-electron] repulsions. 4) Rotate the rear groups on the C2−C3 bond 120∘ counterclockwise from the Newman projection shown, and you'll have it.What is staggered and eclipsed conformations?
Staggered conformations are almost always favored. In an eclipsed conformation the carbons are aligned so that the hydrogens are lined up with each other. This creates steric hindrance between them. In a staggered conformation the atoms are all equally spaced from each other.Which conformation of ethane is more stable?
Staggered conformation of ethane is most stable while eclipsed conformation is least stable because staggered form has the least torsional strain and the eclipsed form has the maximum torsional strain.What is torsional strain?
Torsional strain or eclipsing strain is the increase in potential energy of a molecule due to repulsion between electrons in bonds that do not share an atom. eg: Consider two conformations of ethane: The smallest dihedral angle is 60º in 1; it is 0º in 2. Consequently, torsional strain is greater in 2 than in 1.What are Fichers?
A fissure most commonly occurs after an episode of constipation, but it can happen after an attack of diarrhea. A fissure begins on the surface and usually heals rapidly on its own. Fissures very rarely become infected and they do not become cancerous.What is a chiral molecule?
A chiral molecule is a molecule that is not superimposable on its mirror image. Molecule 2 is not superimposable on its mirror image and, therefore, is chiral. An achiral molecule is a molecule that is superimposable on its mirror image.What is a chiral atom?
Chiral molecules usually contain at least one carbon atom with four nonidentical substituents. Such a carbon atom is called a chiral center (or sometimes a stereogenic center), using organic-speak. When looking at a molecule, look for carbons that are substituted with four different groups.What is the difference between a Fischer projection and a Haworth projection?
While Fischer projections are used for sugars in their open-chain form, Haworth projections are often used to depict sugars in their cyclic forms. The beta diastereomer of the cyclic form of glucose is shown below in three different depictions, with the Haworth projection in the middle.What is meant by Stereoisomer?
Definition of Stereoisomers. Two molecules are described as stereoisomers if they are made of the same atoms connected in the same sequence, but the atoms are positioned differently in space. Optical isomers are molecules which are mirror images of one another.What is absolute configuration in organic chemistry?
An absolute configuration refers to the spatial arrangement of the atoms of a chiral molecular entity (or group) and its stereochemical description e.g. R or S, referring to Rectus, or Sinister, respectively. Absolute configurations are also relevant to characterization of crystals.What are chiral centers?
Chiral centers are tetrahedral atoms (usually carbons) that have four different substituents. Each chiral center in a molecule will be either R or S. As noted above, molecules with a single chiral center are chiral. Molecules with more than one chiral center are usually chiral. The exception are meso-compounds.How many stereoisomers are possible for?
Three stereoisomers are possible: one pair of enantiomers (A and B) and an achiral molecule C, called a “meso compound.” A meso compound is an achiral molecule that nonetheless contains a stereogenic atom.How do you find r and s in Fischer projection?
If the curve goes clockwise, the configuration is R; if the curve goes counterclockwise, the configuration is S. To get the number-four priority substituent at the top of the Fischer projection, you have to use one of the two allowed moves diagramed in the second figure.