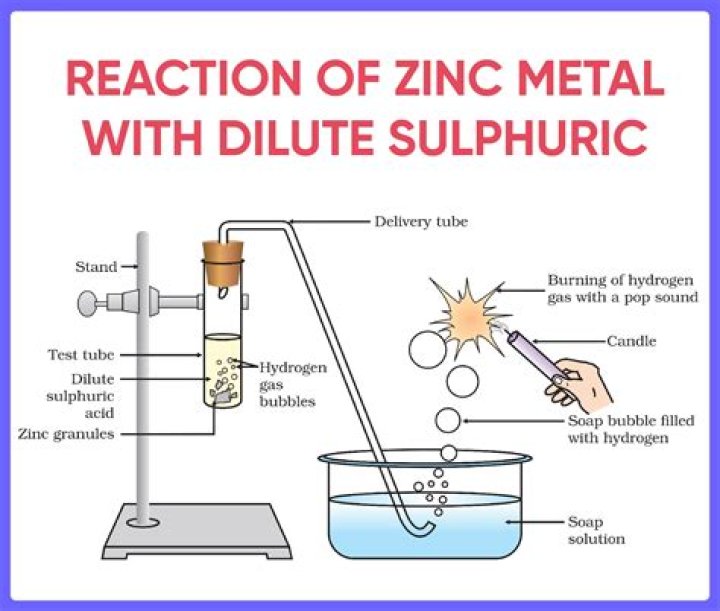
Zinc reacts with sulphuric acid to formzinc sulphate and hydrogen gas is liberated. Zinc +sulphuric acid ——→ zinc sulphate +hydrogen..
Herein, what happens when zinc reacts with sulfuric acid?
Zinc reacts with dilute sulphuric acid toform zinc sulphate and hydrogen gas is evolved. This is asingle displacement reaction of a non-metal by a metal. Thezinc will react with sulphuric acid formingzinc sulphate dissolved in solution and releasing hydrogengas.
Beside above, what does zinc carbonate and Sulphuric acid make? sulfuric acid + zinc carbonate →zinc sulfate + water + carbon dioxide In this type ofreaction an acid reacts with a carbonate to give asalt, water and carbon dioxide.
One may also ask, what forms when sulfuric acid reacts with zinc?
Zinc reacts with sulfuric acid to producehydrogen. The reaction is catalysed by copper. The rate ofreaction can be compared by means of the rate of productionof hydrogen gas bubbles.
Why zinc reacts with sulfuric acid but copper does not?
Note that Copper is a metal which is placed belowHydrogen in the Metallic Reactivity series, hence, it does notreact with any dilute acid to give Hydrogen gas. On theother hand, Zinc is a quite active metal, and is placedabove Hydrogen in the Activity series, so it reacts withdilute acids to form Hydrogen gas.
Related Question Answers
Does Zinc react with sulfuric acid?
Reaction of Zinc with Dilute SulphuricAcid Zinc is more reactive than hydrogen and itdisplaces hydrogen from dilute acids. Zinc reactswith dilute sulphuric acid to form zinc sulphate andhydrogen gas is evolved.What is the formula of dilute Sulphuric acid?
sulfuric acid - chemical formula C2H4 is amineral acid that is soluble in water. Its hydrationreaction is very exothermic, and therefore the acid shouldbe added to the water, because of the densities of the liquids.Sulfuric acid, H2SO4, is a colorless, highly corrosiveoily.What is the Colour of zinc chloride?
white
What is the Colour of zinc?
Zinc is a bluish-silver, lustrous metal thattarnishes in moist air, producing a layer of carbonate. It issomewhat brittle at room temperature but malleable above 100oC. It is a fair conductor of electricity, and burns inair with a bright bluish-green flame producing white clouds of theoxide.How does Zinc react with dilute acids?
Zinc reacts with dilute acid to form salt ofzinc and hydrogen gas . For example : Zinc +Hydrochloric acid _Zinc chloride+hydrogen .What happens when zinc reacts with NaOH?
Zinc reacts with NaOH to form sodium zincate withthe evolution of hydrogen gas. Sodium hydroxide is easilydegraded into sodium carbonate (Na2CO3) in the air because itcontains acidic oxide carbon dioxide (CO2) in the air: 2NaOH + CO2= Na2CO3 + H2O This is also the embodiment of itsbasicity.What is the word equation for zinc and Sulphuric acid?
Zn + H2SO4 → ZnSO4+ H. 2 Zinc react with sulfuric acid to producezinc sulfate and hydrogen. Sulfuric acid - dilutedsolution.What reacts with dilute sulfuric acid?
Dilute sulfuric acid reacts with metals via asingle displacement reaction as with other typical acids,producing hydrogen gas and salts (the metal sulfate). It attacksreactive metals (metals at positions above copper in thereactivity series) such as iron, aluminium, zinc, manganese,magnesium, and nickel. What acids does Zinc react with?
The metal zinc readily reacts withhydrochloric acid to produce hydrogen gas (H2) andzinc chloride (ZnCl2).What happens when Zn reacts with HCl?
It is a single replacement reaction where zincmetal displaces the hydrogen to form hydrogen gas and zincchloride, a salt.Zinc reacts quickly with the acid to formbubbles of hydrogen.Does Zinc react with water?
In what way and in what form does zinc react withwater? This reaction releases hydrogen, whichreacts with oxygen explosively. Zinc salts cause amilky turbidity in water in higher concentrations.Additionally, zinc may add an unwanted flavour towater.What does zinc not react with?
Zinc, as a simple element, reacts withchalcogens, halogens, oxygen, phosphorus, alkalis, acids, ammonium(its salts), and ammonia. Zinc does not enter into areaction with nitrogen, oxygen, boron, carbon andsilicon.Which salt is formed when caco3 reacts with sulfuric acid?
Nitric acid reacts with sodium carbonate toform sodium nitrate, carbon dioxide and water. Sulfuricacid reacts with calcium carbonate to formcalcium sulfate, carbon dioxide and water. Hydrochloric acidreacts with calcium carbonate to form calciumchloride, carbon dioxide and water.Does zinc hydroxide react with hydrochloric acid?
Zinc metal reacts with water to formzinc hydroxide, Zn(OH)2, a white gelatinousprecipitate. This precipitate dissolves upon addition of HClto form zinc chloride, ZnCl2, and water. Write a balancedchemical equation for this reaction between hydrochloricacid and zinc hydroxide.What is produced when an acid reacts with a base?
When an acid and a base are placedtogether, they react to neutralize the acid andbase properties, producing a salt. The H(+) cation of theacid combines with the OH(-) anion of the base toform water. The compound formed by the cation of thebase and the anion of the acid is called asalt.What products are formed when a metal oxide reacts with an acid?
Reactions with metal oxides A salt and water are produced when acidsreact with metal oxides . Metal oxides are bases, because they neutralise acids.What is the product of copper oxide and Sulphuric acid?
Class practical In this experiment an insoluble metal oxide isreacted with a dilute acid to form a soluble salt .Copper(II) oxide , a black solid, and colourlessdilute sulfuric acid react to produce copper(II)sulfate , giving a characteristic blue colour to thesolution.What happens when dilute Sulphuric acid reacts with copper?
Answer: Copper does not react with dilutesulphuric acid. So, no reaction takes place whendilute sulphuric acid is poured on a copper plate.But when concentrated sulphuric acid is poured overcopper plate, effervescence is observed. This happensbecause of formation of hydrogen gas.