Phosphorus pentachloride is the chemical compound with the formula PCl5. It is one of the most important phosphorus chlorides, others being PCl3 and POCl3. It is a colourless, water-sensitive and moisture-sensitive solid, although commercial samples can be yellowish and contaminated with hydrogen chloride..
Moreover, what is the name of PCl5?
Phosphorus(V) chloride Phosphorus pentachloride
Subsequently, question is, what is PCl5 used for? PCl5 is used as an starting material for the synthesis of a variety of inorganic and organic phosphorus compounds used as pesticides, water treatment chemicals, flame retardants, phosphite antioxidants, plasticizers and stabilizers for plastic and elastomers, lube oil and paint additives.
Hereof, what is PCl3 in chemistry?
chemicals. Phosphorus trichloride (PCl3) is used as a chemical intermediate to produce a variety of products which are used in several applications including agricultural products, surfactants and metal extractants, flame retardants, additives for lubricants and stabilizers for plastics.
How is PCl5 formed?
Phosphorus Penta chloride is prepared by passing an excess of dry chlorine into liquid trichloride. Chlorine reacts with phosphorus trichloride and solid phosphorus penta chloride is formed.
Related Question Answers
Is PCl5 poisonous?
PCl5 is a dangerous substance as it reacts violently with water. It is also corrosive when in contact with skin and can be fatal when inhaled.What is the correct name for B2Cl4?
Diboron tetrachloride | B2Cl4 - PubChem.What is structure of PCl5?
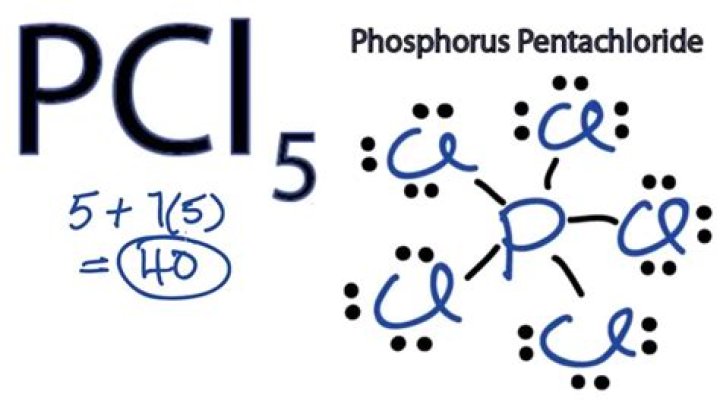
2) Structure of PCl5: It has a trigonal bipyramidal structure, in gaseous and liquid phases. The three equatorial P–Cl bonds are equivalent, while the two axial bonds are longer than equatorial bonds.Is PCl5 solid or liquid?
Yes! Lattice energy is usually the largest factor in determining the stability of an ionic solid. The extra energy gained by the lattice energy more than compensates for the energy needed to transfer a chloride ion from one PCl5 molecule to another. Thus, PCl5 exists as an ionic solid.Is PCl5 acidic?
According to Lewis concept, acid is the substance with empty orbital in valence shell so that it can accept electron pair. PCl5 contain empty d orbital in valence shell. Hence it can acts as Lewis acid.What is the name of cl2o7?
Dichlorine heptoxide is the chemical compound with the formula Cl2O7. This chlorine oxide is the anhydride of perchloric acid.Why PCl5 is very reactive?
In PCl5, in addition to equitorial bonds which lie in the same plane ,there are axial bonds. Since it suffers more repulsion it is longer than the other bonds. Also these are weak bondsand therefore these bonds can break easily . Because of these reasons,PCl5 is very reactive.What is the formula for carbon Pentachloride?
Carbon tetrachloride, also known by many other names (such as tetrachloromethane, also recognised by the IUPAC, carbon tet in the cleaning industry, Halon-104 in firefighting, and Refrigerant-10 in HVACR) is an organic compound with the chemical formula CCl4.Is PCl3 a Lewis acid?
Lewis acids are electron acceptors. Phosphorous has empty d orbitals in its valence shell to accept electrons and hence acts as Lewis acid. IN PCL3 , PHOSPHORUS HAVE A FIVE VALENCE ELECTRONS IN WHICH THREE ELECTRONS ARE SHARED WITH THREE CHLORINE ATOMS DUE TO WHICH THREE COVALENT BONDS ARE FORMED.Is PCl3 explosive?
Phosphorus trichloride is a chemical compound of phosphorus and chlorine, having the chemical formula PCl3. It is a toxic and volatile liquid which reacts violently with water to release HCl gas.How is PCl3 made?
Phosphorus trichloride is prepared industrially by the reaction of chlorine with a refluxing solution of white phosphorus in phosphorus trichloride, with continuous removal of PCl3 as it is formed. In the laboratory it may be more convenient to use the less toxic red phosphorus.Is PCl3 a gas?
Phosphorus trichloride (phosphorus chloride, PCl3) is a colorless fuming liquid that reacts violently with water to liberate phosphoric acid and hydrogen chloride (HCL) gas. PCl3 is a strong oxidizer and will readily react with many organic compounds.What is the chemical name of POCl3?
phosphorus oxychloride
Is POCl3 an acid?
POCl3 will not bioaccumulate but is not biodegradable. POCl3 in higher concentrations can be harmful to aquatic life due to formation of acids from the hydrolysis of POCl3. The vapors hydrolyze to hydrochloric and phosphoric acids from reaction with humidity in the air.What is the full form of PCl3?
Phosphorus trichloride (phosphorus chloride, PCl3) is a colorless fuming liquid that reacts violently with water to liberate phosphoric acid and hydrogen chloride (HCL) gas.Is PCl3 soluble in water?
Water
How many total electrons are in phosphorus trichloride?
PCL3 Electron Geometry Now we all are clear that the Phosphorus has 5 valence electrons and the chlorine has 7 valence electrons. There are three chlorines, and so the seven must be multiplied with three, which gives the output of 21. Now, this 21 should be added in 5 – the valence electrons of Phosphorus.Why PCl5 is a good chlorinating agent?
(a) PCl5 has a trigonal bipyramidal shape in which P—Cl (axial) bonds are slightly larger than the P—Cl (equatorial) bonds. This makes the molecule, rather, unstable. It dissociates to lose two chlorine atoms involved in the axial bonds and thus, acts as a chlorinating agent.What is the hybridization of PCl5?
The hybridization of P in PCl5 is sp3d. The P atom needs five orbitals to form the five P-Cl bonds . It has a 3s and three 3p orbitals, so it must use one of its 3d orbitals to form the fifth bond.These orbitals are hybridized to form five sp3d orbitals and thus form a trigonal bipyramid.