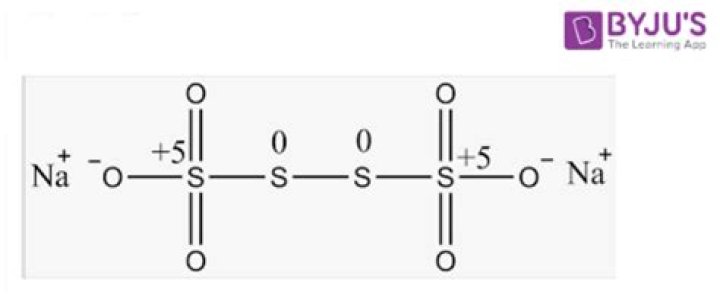
The tetrathionate anion,S. 4O 2− 6, is a sulfur oxoanion derived from the compound tetrathionic acid, H2S4O6. Two of the sulfur atoms present in the ion are in oxidation state 0 and two are in oxidation state +5. Alternatively, the compound can be viewed as the adduct resulting from the binding of S 2−.
In this regard, what is the oxidation number of Na2S4O6?
his compound must have sulfur atoms with mixed oxidation states. Since it is normal for sulfur to have oxidation states of -2, 0, +2, +4, and +6, it is most likely that there are three sulfurs with a +2 oxidation state and one sulfur that is +4.
Secondly, what is the oxidation number of Sulphur in N 82 S o4? In thiosulphate, two sulphurs have oxidation state of -2 and +6. (as per suggestions). Sulphur bonded to three oxygen is considered to have +6 (Sulphur A) and other sulphur has -2 (Sulphur B).
Beside this, what is the structure of Na2S4O6?
Sodium tetrathionate is a salt of sodium and tetrathionate with the formula Na2S4O6xH2O. The salt normally is obtained as the dihydrate (x = 2). It is a colorless, water-soluble solid. It is one member of the polythionates, which have the formula [Sn(SO3)2]2-.
How do you find the oxidation number of Na2SO4?
For example, Na2SO4 has two sodium (Na) atoms with an oxidation charge of plus 1 each and an overall oxidation value of plus 2. Add together the oxidation values for all known species. Subtract the summed value from the overall charge of the compound. For example, Na2SO4, Na2 yields 2 (plus 1) + 4(minus 2) = minus 6.
Related Question Answers
How do you get the oxidation number?
1 Answer - The oxidation number of a free element is always 0.
- The oxidation number of a monatomic ion equals the charge of the ion.
- The oxidation number of H is +1, but it is -1 in when combined with less electronegative elements.
- The oxidation number of O in compounds is usually -2, but it is -1 in peroxides.
What is the oxidation number of na2so3?
Therefore, the oxidation number of sulfur is +4 (it lost four electrons to oxygen) and the oxidation numbers for our compound is as follows: Na +1; S +4; O -2. You can also do this algebraically by setting up an equation equal to zero.What is disproportionation reaction give example?
A disproportionation reaction is when a multiatomic species whose pertinent element has a specific oxidation state gets oxidized and reduced in two separate half-reactions, yielding two other products containing the same pertinent element. EXAMPLE: MANGANESE OXIDES. A convenient example is Mn2O3 becoming Mn2+ and MnO2What happens to the oxidation number of an element during oxidation?
When oxidation occurs, an element loses electrons and its oxidation number increases (becomes more positive). When reduction occurs, an element gains electrons and its oxidation number decreases or is reduced (becomes more negative).What is the oxidation number of N in nh4no3?
The oxidation of nitrogen in NH4+ is -3. nitrogen is surrounded by the four hydrogen atoms have +1 and there are four of them. And net charge on the molecule is +1.Can oxidation state fractional?
Fractional Values for Oxidation States The oxidation states are usually in whole numbers, but in some cases, they are in fractional numbers. Consider Fe3O4. In most cases, atoms of the same element in a given compound have the same oxidation states, but each atom can have a different state than the other.What is the oxidation number of Sulphur in h2so4?
Thus oxidation number of sulfur in H2SO4 is +6.What is the oxidation state of sulfur in cuso4?
The oxidation number of the sulfur atom in the SO42- ion must be +6, for example, because the sum of the oxidation numbers of the atoms in this ion must equal -2.What is the name of s4o62?
Tetrathionate. Description: Tetrathionate(2-) is a tetrathionate ion and a sulfur oxide.What does sodium thiosulfate do?
Sodium thiosulfate, also spelled sodium thiosulphate, is used as a medication to treat cyanide poisoning, pityriasis versicolor, and to decrease side effects from cisplatin. For cyanide poisoning it is often used after the medication sodium nitrite and typically only recommended for severe cases.What are the oxidation numbers of chlorine in CA OCl CL?
In bleaching powder, Ca(OCl)Cl, the two Cl atoms are in different oxidation states i.e., one Cl– having oxidation number of –1 and the other as OCl– having oxidation number of +1. x + 3 (– 2) = –1 or x = + 5. In Fe3O4, Fe atoms are in two different oxidation states.What is the oxidation number of chromium in K2Cr2O7?
So in K2Cr2O7 , the oxidation number of Cr is +6.What is the oxidation state of Sulphur in so2?
+4
Which of the following is a reduction oxidation redox reaction?
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. An oxidation-reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron.What is the oxidation number of nitrogen in CA no3 2?
Ca(NO3)2 can be broken into two ions Ca2+ and NO3-. Using oxidation rules, nitrogen has an oxidation number of x + 3 x -2 = -1; N = +5.What is the charge of na2so4?
Sodium Sulfate is an ionic compound formed by two ions, Sodium Na+ and Sulfate SO−24 . In order for these two polyatomic ions to bond the charges must be equal and opposite. Therefore, it will take two +1 sodium ions to balance the one -2 sulfate ion.What is the charge of so4?
We would need 32 valence electrons for 4 full outer shells(8x4) but only have 30. Thus the charge is 2- for the ion.What is the oxidation number of nahso4?
The oxidation state of sulfur in sodium hydrogen sulfate is +6 .What is the oxidation number of phosphorus in h3po4?
We know from part (b) that the oxidation number of the phosphorus atoms in H3PO4 is +5.