What is metallic property in chemistry?
.
Correspondingly, what does metallic mean in chemistry?
Metallic character refers to the level of reactivity of a metal. Metals tend to lose electrons in chemical reactions, as indicated by their low ionization energies. Within a compound, metal atoms have relatively low attraction for electrons, as indicated by their low electronegativities.
Likewise, what are metallic properties examples? A metal can refer to an element, compound, or alloy that is a good conductor of both electricity and heat. Example metals include gold, sodium, copper, iron, and many other elements. Metals are usually malleable, ductile, and shiny.
Correspondingly, how do you know if an element is metallic?
You can predict the metallic character of an element using the periodic table. Metallic character increases as you move down a group (column) of the periodic table. Metallic character decreases as you move from left to right across a period (row) of the periodic table.
What is metallic property in periodic table?
Metallic character refers to the level of reactivity of a metal. Non-metallic character relates to the tendency to accept electrons during chemical reactions. Metallic tendency increases going down a group. Non-metallic tendency increases going from left to right across the periodic table.
Related Question AnswersHow is metallic character defined?
Metallic Character Definition Metallic character describes the set of chemical properties that are associated with the elements classified as metals in the periodic table. Metallic character depends on the ability of an element to lose its outer valence electrons.Which non metal is most reactive?
fluorineWhat is non metal in science?
Non-metals are the elements in groups 14-16 of the periodic table. As opposed to metals, non-metallic elements are very brittle, and cannot be rolled into wires or pounded into sheets. The non-metals exist in two of the three states of matter at room temperature: gases (such as oxygen) and solids (such as carbon).What is metallic made of?
Metallic fibers are manufactured fibers composed of metal, metallic alloys, plastic-coated metal, metal-coated plastic, or a core completely covered by metal. Having their origin in textile and clothing applications, gold and silver fibers have been used since ancient times as yarns for fabric decoration.Which group is most metallic?
Explanation: Metallic character increases form right to left across a period on the periodic table, and from top to bottom down a group. The alkali metals in group 1 are the most active metals, and cesium is the last element in the group for which we have experimental data.What is a metal in science?
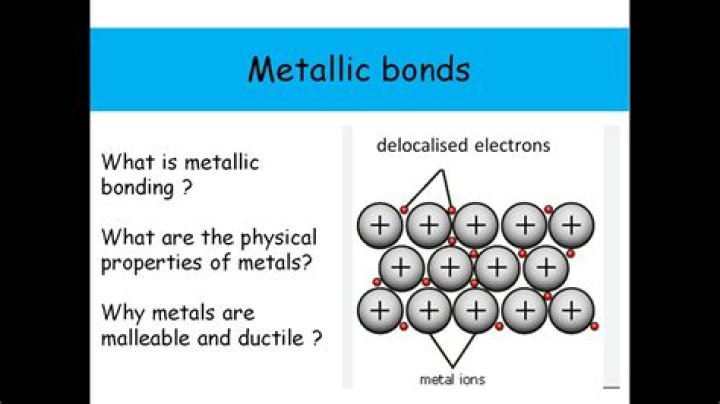
In chemistry, a metal is an element that readily forms positive ions (cations) and has metallic bonds. Metals are sometimes described as a lattice of positive ions surrounded by a cloud of delocalized electrons.Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.Who discovered metal?
People first began making things from metal over 9000 years ago, when they discovered how to get copper from its []ore. They then learned how to make a harder alloy, bronze, by adding tin to the copper. About 3000 years ago, they discovered iron.What is the heaviest element?
uraniumWhat are the 22 non metals?
In modern periodic table there are 22 non-metals in which there are 11 gases, 1 liquid and 10 solid. Bromine occurs in the state of liquid and hydrogen, nitrogen, oxygen, chlorine etc are found in gaseous forms. But carbon, sulphur, phosphorous, iodine etc solid non-metals.How do you tell if an element is a nonmetal?
Elements to the left of the line are considered metals. Elements just to the right of the line exhibit properties of both metals and nonmetals and are termed metalloids or semimetals. Elements to the far right of the periodic table are nonmetals. The exception is hydrogen (H), the first element on the periodic table.What is the least metallic element?
This makes sense, given that the metals are on the left and downwards generally. Therefore, the least metallic elements are the opposite: the right and uppermost elements on the periodic table. Thus, the elements Helium, Neon, Fluorine, and Oxygen are logically the least metallic.How can we find Valency?
The valency of an atom is equal to the number of electrons in the outer shell if that number is four or less. Otherwise, the valency is equal to eight minus the number of electrons in the outer shell. Once you know the number of electrons, you can easily calculate the valency.Is PA a metal or nonmetal?
Phosphorous is a nonmetal. Majority of elements are metals. If we consider up to Uranium (92, last naturally occurring element) 17 elements are classified as nonmetals, 7 are classified as metalloids, and the rest are metals.How do you know if an element is malleable?
If malleable, a material may be flattened into thin sheets by hammering or rolling. Malleable materials can be flattened into metal leaf. One well-known type of metal leaf is gold leaf. Many metals with high malleability also have high ductility.Which element has the highest ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).Which is not a property of metals?
Non-metals are poor conductors of heat and electricity except Graphite. They do not possess metallic luster. Non-metals are not sonorous, they do not produce a ringing sound. Non-metals possess no malleability.What are the types of metals?
18 Different Types of Metal (Facts and Uses)- Steel. Carbon Steel. Alloy Steel. Stainless Steel.
- Iron (Wrought or Cast)
- Aluminum.
- Magnesium.
- Copper.
- Brass.
- Bronze.
- Zinc.



