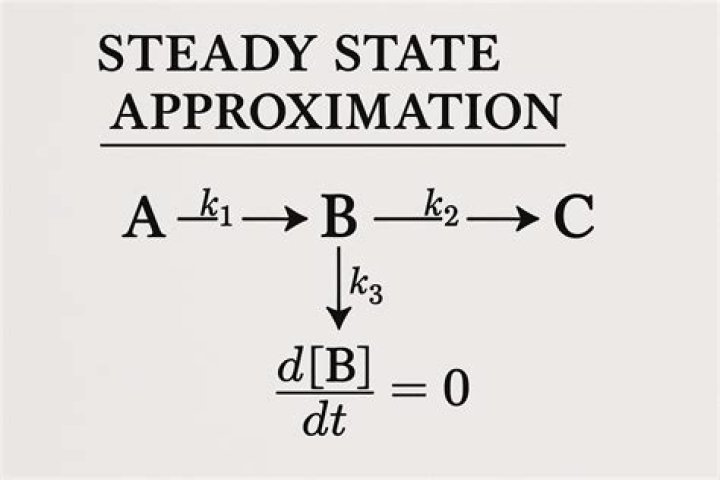
The steady-state approximation is a method used to derive a rate law. The method is based on the assumption that one intermediate in the reaction mechanism is consumed as quickly as it is generated. Its concentration remains the same in a duration of the reaction. Definition: Intermediates..
Hereof, how do you use steady state approximation?
The steady state approximation is applies to a consecutive reaction with a slow first step and a fast second step (k1≪k2). If the first step is very slow in comparison to the second step, there is no accumulation of intermediate product, such as product B in the above example.
Also Know, what is the difference between steady state and pre steady state? pre steady state kinetics would involve the components comprising a chemical reaction, such as hydrogen and oxygen; steady state kinetics would involve the results of that chemical reaction, water plus energy, as in this example of an exothermic reaction.
Herein, what does steady state mean in chemistry?
In chemistry, a steady state is a situation in which all state variables are constant in spite of ongoing processes that strive to change them. For an entire system to be at steady state, i.e. for all state variables of a system to be constant, there must be a flow through the system (compare mass balance).
What is the steady state equilibrium?
Steady State. A system that is in a steady state remains constant over time, but that constant state requires continual work. This condition is also referred to as a system in dynamic equilibrium. A system in a steady state has a higher level of energy than its surroundings.
Related Question Answers
What happens in a steady state?
A steady state flow process requires conditions at all points in an apparatus remain constant as time changes. There must be no accumulation of mass or energy over the time period of interest. The same mass flow rate will remain constant in the flow path through each element of the system.Why is the steady state approximation useful?
The steady-state approximation is a method used to derive a rate law. The method is based on the assumption that one intermediate in the reaction mechanism is consumed as quickly as it is generated. Its concentration remains the same in a duration of the reaction.What is the difference between steady state and equilibrium?
Yes, equilibrium and steady-state are distinct concepts. A reaction is at equilibrium if reactants and products are both present, the forward and reverse rates are equal and the concentrations don't change over time. Steady-state implies a system that is not at equilibrium (entropy increases).What is dynamic steady state?
Steady State. A system that is in a steady state remains constant over time, but that constant state requires continual work. This condition is also referred to as a system in dynamic equilibrium. A system in a steady state has a higher level of energy than its surroundings.What is steady state exercise?
Steady state exercise is activity that achieves a balance between the energy required by working muscles and the rate of oxygen and delivery for aerobic ATP production. It has to beat a steady rate, or it will change from being steady state exercise to being anaerobic or high level aerobic exercise.What is steady state in macroeconomics?
Summary. A steady state economy is an economy of stable or mildly fluctuating size. The term typically refers to a national economy, but it can also be applied to a local, regional, or global economy. An economy can reach a steady state after a period of growth or after a period of downsizing or degrowth.What is a steady state flow?
Steady-state flow refers to the condition where the fluid properties at any single point in the system do not change over time. These fluid properties include temperature, pressure, and velocity. One of the most significant properties that is constant in a steady-state flow system is the system mass flow rate.What do you mean by steady state response?
(Electrical engineering: Computing and control) A steady-state response is the behavior of a circuit after a long time when steady conditions have been reached after an external excitation.What is steady flow?
A steady flow is the one in which the quantity of liquid flowing per second through any section, is constant. This is the definition for the ideal case. True steady flow is present only in Laminar flow. In turbulent flow, there are continual fluctuations in velocity. Pressure also fluctuate at every point.What is steady state kinetics?
Steady-state kinetics applies whenever the concentration of the substrate is well above that of the enzyme, so that the rate of change of substrate concentration greatly exceeds the rate of change of the concentration of any enzyme form.What makes conditions steady state with regards to kinetics experiments?
The steady-state approximation is a method used to derive a rate law. The method is based on the assumption that one intermediate in the reaction mechanism is consumed as quickly as it is generated. Its concentration remains the same in a duration of the reaction. Thus, the system has reached a steady-state.What is the steady state assumption in enzyme kinetics?
Steady state occurs when the rate of formation and breakdown of the intermediate are equal. The steady state assumption relies on the fact that both the formation of the intermediate from reactants and the formation of products from the intermediate have rates much higher than their corresponding reverse reactions.What is v0 in enzyme kinetics?
Michaelis-Menten Kinetics. A plot of the reaction velocity (V0) as a function of the substrate concentration [S] for an enzyme that obeys Michaelis-Menten kinetics shows that the maximal velocity (Vmax) is approached asymptotically.What is single turnover kinetics?
The kinetics of an enzyme-catalysed reaction in conditions under which formation of the enzyme-substrate complex can be measured. Such conditions are required for the detection and analysis of transient enzyme-substrate From: single-turnover kinetics in Oxford Dictionary of Biochemistry and Molecular Biology »What burst kinetics?
Burst kinetics is a form of enzyme kinetics. Upon adding enzyme to substrate, a large initial velocity is exhibited that levels off once all enzymes have been saturated.What is an example of steady state?
The definition of a steady-state is an unchanging condition, system or physical process that remains the same even after transformation or change. When you have a chemical mix that has certain properties, and the mix retains those properties even after you add a change-agent, this is an example of a steady-state.What is the steady state value?
The term Steady state gain comes up when your input function is the unit step function u(t)=1. If your input is the unit step function, then the gain is the system's value at steady state, t=∞. The steady state value is also called the final value. So if your transfer function is H(z)=Y(z)X(z)=.