What is meant by free radical mechanism?
.
Just so, what is a radical mechanism?
Unlike the large majority of reactions that you will see in your organic chemistry course, radical mechanisms require that fishhook curly arrows that represent the motion of a single electron are used.
One may also ask, what makes a free radical? Oxidative stress occurs when an oxygen molecule splits into single atoms with unpaired electrons, which are called free radicals. Electrons like to be in pairs, so these atoms, called free radicals, scavenge the body to seek out other electrons so they can become a pair. This causes damage to cells, proteins and DNA.
Secondly, what is a free radical example?
A notable example of a free radical is the hydroxyl radical (HO•), a molecule that is one hydrogen atom short of a water molecule and thus has one bond "dangling" from the oxygen.
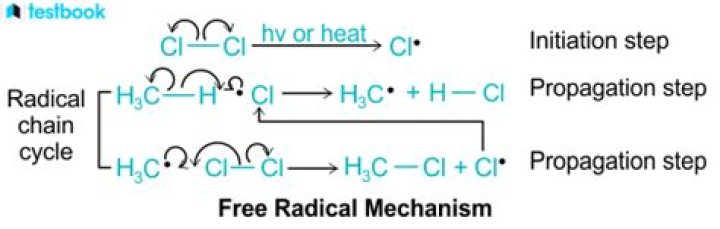
What are the three steps in a radical mechanism?
Radical chain reactions have three distinct phases: initiation, propagation, and termination.
- The initiation phase describes the step that initially creates a radical species.
- The propagation phase describes the 'chain' part of chain reactions.
Is oxygen a free radical?
A free radical is any species capable of independent existence containing one or more unpaired electrons [1]. The oxygen free radicals include superoxide anion radical (O2·–), singlet oxygen (1O2), hydroxyl radical (·OH) and perhydroxyl radical (HO2·) and are termed collectively the 'reactive oxygen species' (ROS).How do you find radical stability?
If the internal energy of the radical is low, the radical is stable. It will have little tendency to react further. Free radicals have only 7 electrons in their valence shell. They are higher in energy than atoms with 8 valence electrons.How are radicals formed?
Free radicals form when one of these weak bonds between electrons is broken and an uneven number of electrons remain. This means the electron is unpaired, making it chemically reactive. It will now try and steal an electron from a neighboring molecule to stabilize itself.How do you name radicals?
A radical derived formally by the removal of one or two hydrogen atoms from an amine, imine, or amide characteristic group may be named by adding a suffix "-aminyl", "-iminyl", or "-amidyl", to the name of the parent hydride for monovalent radicals and as a substituted nitrene for bivalent radicals.What type of reaction is free radical substitution?
In organic chemistry, a radical-substitution reaction is a substitution reaction involving free radicals as a reactive intermediate. The reaction always involves at least two steps, and possibly a third. In the first step called initiation (2,3) a free radical is created by homolysis.What is carbon free radical?
A free radical is an atom or a group of atoms with an odd number of electrons. An organic free radical is a free radical form of carbon with three bonds and a single, unpaired electron. A free radical can react with another free radical, but more often it reacts with a stable, evenly paired molecule.What is a radical chain reaction?
Radical chain reaction (free radical chain reaction): A radical reaction mechanism in which one or more steps are repeated many times, until the chain (the sequence of repeated steps) is terminated, or until one of the reactants is depleted.What are basic radicals?
Basic radical is an ion coming from a base. It is a positively charged chemical species; thus we name it as the cation. Moreover, it is a portion of an inorganic salt. This ion forms as a result of the removal of a hydroxide ion from a base.What are radicals and examples?
Radical (chemistry)- In chemistry, a radical is an atom, molecule, or ion that has an unpaired valence electron.
- A notable example of a radical is the hydroxyl radical (HO•), a molecule that has one unpaired electron on the oxygen atom.
- Radicals may be generated in a number of ways, but typical methods involve redox reactions.



