What is mass percent of a solution?
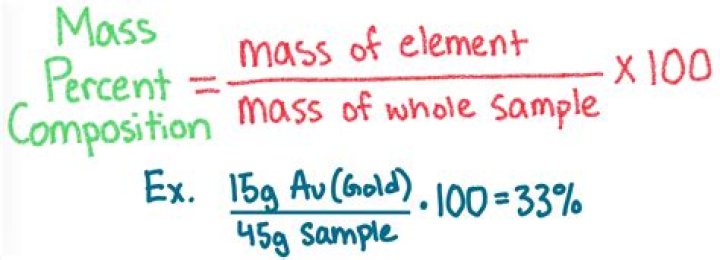
.
In this way, what is meant by mass percentage of solution?
Mass percentage of a solution is a method to express the concentration or the amount of a component present in a given quantity of solution. The mass percentage of a component in a solution is defined as the mass of solute present per 100 grams of the solution and is given by the following relation.
Likewise, what is a percent solution? A percentage solution is an amount or volume of chemical or compound per 100 mL of a solution. It is a relative expression of solute to solvent: X amount/100 ml = X% Percentage solutions are a convenient and easy way to record solution concentrations.
In this regard, how do you calculate mass percent concentration?
Percent by mass (m/m) is the mass of solute divided by the total mass of the solution, multiplied by 100 %.
- Percent by mass = mass of solutetotal mass of solution × 100 %
- mass of glucosetotal mass of solution×100%=26.5g500g × 100 % = 5.30 %
- Percent by volume = volume of solutetotal volume of solution × 100 %
What is the formula for the mass?
Part 2 Newton's second law of motion states that force equals mass times acceleration: F = ma. If you know the net force on the object, and it's acceleration, you can rearrange this formula to find the mass: m = F / a. Force is measured in N (newton), which you can also write as (kg * m)/ s2.
Related Question AnswersWhat is the formula of mass percentage?
Calculate the mass percent. Divide the mass of the element by the total mass of the compound and multiply by 100. This will give you the mass percent of the element. Example 1: mass percent = (2.01588/18.01528) x 100 = 0.11189 x 100 = 11.18%. Thus, the mass percent of Hydrogen atoms in a water molecule is 11.18%.How do you make a 5% solution?
Dilute the compound with the necessary amount of solvents. For example: Mix 500 mL of water and 25 g of NaCl to make a 5% solution. Remember, if you're diluting a liquid compound, you must subtract out the volume of liquid being added from the final volume: 500 mL – 25 mL = 475 mL of water.What is the mass percent of a solution?
To calculate the mass percent or weight percent of a solution, you must divide the mass of the solute by the mass of the solution (both the solute and the solvent together) and then multiply by 100 to change it into percent.What is Molality formula?
The formula for molality is m = moles of solute / kilograms of solvent. In problem solving involving molality, we sometimes need to use additional formulas to get to the final answer. One formula we need to be aware of is the formula for density, which is d = m / v, where d is density, m is mass and v is volume.How do you solve for mass of solute?
Mass of Solute and Molar Mass The number of moles of solute = mass of solute ÷ molar mass of solute, where mass is measured in grams and molar mass (defined as the mass of one mole of a substance in grams) is measured in g/mol. If you don't know the mass of the solute, weigh it on a scale and record the value.How do you make a 1 percent solution?
The mass of a solute that is needed in order to make a 1% solution is 1% of the mass of pure water of the desired final volume. Examples of 100% solutions are 1000 grams in 1000 milliliters or 1 gram in 1 milliliter.How can calculate percentage?
1. How to calculate percentage of a number. Use the percentage formula: P% * X = Y- Convert the problem to an equation using the percentage formula: P% * X = Y.
- P is 10%, X is 150, so the equation is 10% * 150 = Y.
- Convert 10% to a decimal by removing the percent sign and dividing by 100: 10/100 = 0.10.
Whats is a solution?
A solution is a homogeneous type of mixture of two or more substances. A solution has two parts: a solute and a solvent. The solute is the substance that dissolves, and the solvent is the majority of the solution. Solutions can exist in different phases - solid, liquid, and gas.How do I calculate moles?
- Start with the number of grams of each element, given in the problem.
- Convert the mass of each element to moles using the molar mass from the periodic table.
- Divide each mole value by the smallest number of moles calculated.
- Round to the nearest whole number. This is the mole ratio of the elements and is.
How do you find Percent Composition?
Percent Composition- Find the molar mass of all the elements in the compound in grams per mole.
- Find the molecular mass of the entire compound.
- Divide the component's molar mass by the entire molecular mass.
- You will now have a number between 0 and 1. Multiply it by 100% to get percent composition.
How do u calculate percent error?
Steps to Calculate the Percent Error- Subtract the accepted value from the experimental value.
- Take the absolute value of step 1.
- Divide that answer by the accepted value.
- Multiply that answer by 100 and add the % symbol to express the answer as a percentage.