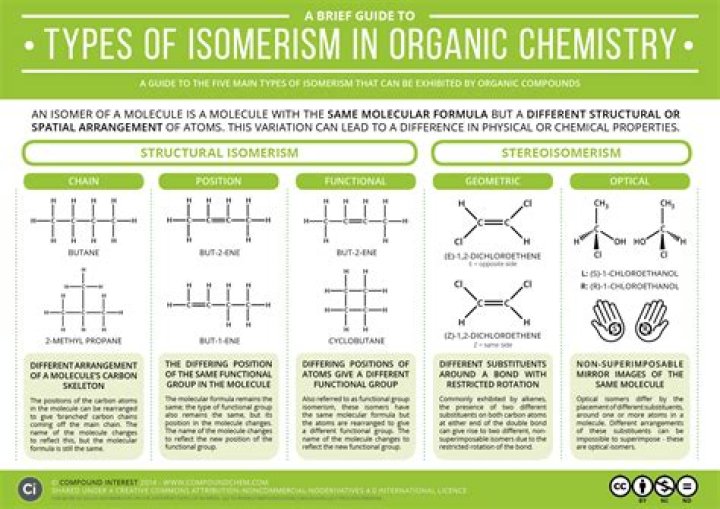
In organic chemistry, isomers are molecules with the same molecular formula (i.e. the same number of atoms of each element), but different structural or spatial arrangements of the atoms within the molecule..
In respect to this, what is isomerism and example?
Isomers are molecules that have the same molecular formula, but have a different arrangement of the atoms in space. That excludes any different arrangements which are simply due to the molecule rotating as a whole, or rotating about particular bonds. For example, both of the following are the same molecule.
Subsequently, question is, what are the 3 types of isomers? There are three types of structural isomers: chain isomers, functional group isomers and positional isomers. Chain isomers have the same molecular formula but different arrangements or branches. Functional group isomers have the same formula but different functional groups.
Hereof, what is polymerisation isomerism?
Polymerization isomers, according to our textbook, are. those complexes that have the same empirical formula, but differ in molecular mass by integral multiples of empirical mass.
What are the types of isomerism in complexes?
Isomers that contain the same number of atoms of each kind but differ in which atoms are bonded to one another are called structural isomers, which differ in structure or bond type. For inorganic complexes, there are three types of structural isomers: ionization, coordination and linkage.
Related Question Answers
What is called isomerism?
Isomerism is the phenomenon whereby certain compounds, with the same molecular formula, exist in different forms owing to their different organisations of atoms. The concept of isomerism illustrates the fundamental importance of molecular structure and shape in organic chemistry.What are different types of isomers?
There are two broad types of isomers: constitutional and stereoisomers. Constitutional isomers differ in bonding and connectivity. Stereoisomers differ in 3D orientation. Enantiomers and diastereomers are two sub-types of stereoisomers.Why are isomers important?
They are important because two isomers can have the same chemical formula, but have different chemical structures. The structure contributes to the properties of the molecule.What is isomerism in chemistry?
In organic chemistry, isomers are molecules with the same molecular formula (i.e. the same number of atoms of each element), but different structural or spatial arrangements of the atoms within the molecule.How do you determine isomers?
Constitutional isomers are compounds that have the same molecular formula and different connectivity. To determine whether two molecules are constitutional isomers, just count the number of each atom in both molecules and see how the atoms are arranged.Is glucose and fructose isomers?
Glucose, galactose, and fructose are monosaccharide isomers, which means they all have the same chemical formula but differ structurally and chemically.What compounds are isomers?
Just as with metal complexes, compounds that have the same molecular formula but different arrangements of atoms are called isomers. In this section, we describe various types of isomers, beginning with those whose three-dimensional structures differ only as the result of rotation about a C–C bond.What are Metamers?
Compounds having the same molecular formula but different number of carbon atoms ( alkyl groups) on either side of functional group ( i.e., -O-,-S-, -NH-, -C(=O)-) are called metamers and the phenomenon is called metamerism.What is stereoisomers in organic chemistry?
Stereoisomerism is the arrangement of atoms in molecules whose connectivity remains the same but their arrangement in space is different in each isomer. The two main types of stereoisomerism are: DiaStereomerism (including 'cis-trans isomerism') Optical Isomerism (also known as 'enantiomerism' and 'chirality')What are stereoregular polymers?
polymers are referred to as stereoregular—that is, polymers having an ordered arrangement of pendant groups along the chain. A polymer with a random orientation of groups is said to be atactic.What is optical isomerism?
Optical isomers are two compounds which contain the same number and kinds of atoms, and bonds (i.e., the connectivity between atoms is the same), and different spatial arrangements of the atoms, but which have non-superimposable mirror images. Each non-superimposable mirror image structure is called an enantiomer.Are fac and mer isomers optically active?
The fac isomer is where the ligands are on the same plane and there exists a 90-90-90 degree bond angle between the 3 same ligands. 4) A dextrorotatory optical isomer is a isomer that can rotate light in the right direction. Thus, this molecule is optically active because it has optical isomers.How do you find geometrical isomers in coordination compounds?
Three ligands are in the equatorial plane and the other two are at the axial positions, trans to each other. We can count the geometrical isomers by counting all the possible trans ligand pairs.Which complex compound will give four isomers?
ANSWER:- [Fe(PPh3)3NH3ClBr]Cl can give two optical and two geometrical isomers. total 4 isomer. [Fe(PPh3)3NH3ClBr]Cl can give two optical and two geometrical isomers.Which isomerism is present in coordination compounds?
As with other compounds, there are several kinds of coordination complex isomers. Stereoisomers occur when the ligands have the same bonds, but the bonds are in different orientations relative to one another. Structural isomerism occurs when the bonds are different.What do you understand by isomerism in coordination compounds also explain its types?
Isomerism in Coordination Compounds. Compounds that have the same chemical formula but different structural arrangements are called isomers. Due to their complicated formulae of many coordination compounds, the variety of bond types and the number of shapes possible, many different types of isomerism occur.What is ionization isomerism in coordination compound?
Ionization isomers are identical except for a ligand has exchanged places with an anion or neutral molecule that was originally outside the coordination complex. The central ion and the other ligands are identical. For example, an octahedral isomer will have five ligands that are identical, but the sixth will differ.What is a chiral molecule?
Definitions: Chiral. A molecule is chiral if it is not superimposable on its mirror image. Most chiral molecules can be identified by their lack of a plane of symmetry or a center of symmetry. Your hand is a chiral object, as it does not have either of these types of symmetry.What do you mean by structural isomers?
Structural Isomers are molecules which have the same molecular formula but have different connectivities (The Order They Are Put Together). Alkanes can be very simple examples of this. With the structural formula C4H10 there are two different isomers possible.