Description: Iodite is a monovalent inorganic anion obtained by deprotonation of iodous acid. It is an iodine oxoanion and a monovalent inorganic anion..
Just so, what is the formula for Iodite?
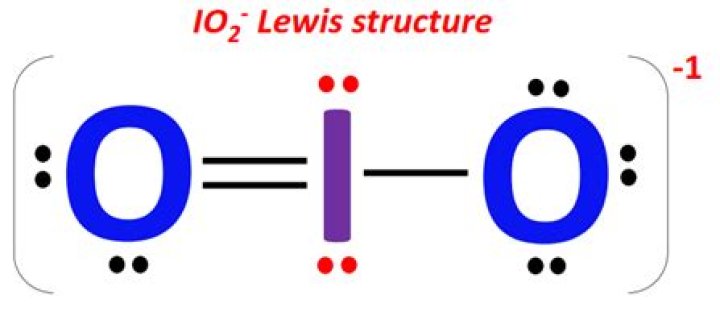
Iodite is a polyatomic ion. It has the formula IO2 -1. The iodine atom is the central atom in the molecule with one oxygen atom on either side of it.
what is c2h3o2? Acetate is a compound derived from acetic acid. When the hydroxyl group is replaced by an alkoxy group, an acetate ester is formed. The molecular formula for acetate is C2H3O2-. The solubility of acetate is related specifically to its form as a salt.
Just so, what is OI in chemistry?
The Iodite ion, or iodine dioxide anion, is the halite with the chemical formula IO − . Within the ion the Iodine exists in the oxidation state of +3.
What is Iodite used for?
Calcium iodate is an oxidant added to lotions and ointments as an antiseptic and deodorant. It is also used in the food industry in animal feed, and in the manufacture of disinfectants. Iodite is an anion composed of iodine and oxygen, with the chemical formula IO2-.
Related Question Answers
What is the name for io2?
iodite. dioxidoiodate(1-) CHEBI:29230. IO2(-) [IO2](-)What is bro4?
In chemistry, the perbromate ion is the anion having the chemical formula BrO − 4. It is an oxyanion of bromine, the conjugate base of perbromic acid, in which bromine has the oxidation state +7.What is the symbol of iodide?
Iodine is a chemical element with the symbol I and atomic number 53. The heaviest of the stable halogens, it exists as a lustrous, purple-black non-metallic solid at standard conditions that melts to form a deep violet liquid at 114 degrees Celsius, and boils to a violet gas at 184 degrees Celsius.What is the formula for periodate?
Periodate is an ion. Its chemical formula is either IO4- (metaperiodate) or IO65- (orthoperiodate). They are the salts of periodic acid.What is the name for ch3co2?
acetate. (redirected from CH3CO2-)How do you determine molar mass?
Key Points - The molar mass is the mass of a given chemical element or chemical compound (g) divided by the amount of substance (mol).
- The molar mass of a compound can be calculated by adding the standard atomic masses (in g/mol) of the constituent atoms.
How do you name polyatomic ions?
Rule 1. The cation is written first in the name; the anion is written second in the name. Rule 2. When the formula unit contains two or more of the same polyatomic ion, that ion is written in parentheses with the subscript written outside the parentheses.Is no3 a molecule?
The nitrate ion is a polyatomic anion with the empirical formula NO3- and a molecular mass of 62. 01 daltons; it consists of one central nitrogen atom surrounded by three identical oxygen atoms in a trigonal planar arrangement.Is HIO a weak acid?
Hypoiodous acid (HIO) is a weak acid that dissociates in water as follows: HIO(aq) + H2O(l) equilibrium reaction arrow H3O+(aq) + IO−(aq). A 0.15 M solution of hypoiodous acid has a pH of 5.66.What is Hypoiodite reagent?
Hypoiodite is an ion. Its chemical formula is IO-. It is very unstable. It easily disproportionates to iodide and iodate. It is made by reacting iodine with a base.What is the oxidation number of iodine in Iodous acid?
Iodic acid, HIO3. It is a white water-soluble solid. Its robustness contrasts with the instability of chloric acid and bromic acid. Iodic acid features iodine in the oxidation state +5 and is one of the most stable oxo-acids of the halogens.Is c2h3o2 an acid or base?
C2H3O2– on the other can accept a proton so C2H3O2– is a base but its a conjugate base to acid HC2H3O2 since it is generatd by losing a proton from HC2H3O2 . Similary, H3O+ is an acid as it can now donate a proton but it is a conjugate acid to the base H2O as it has been generated by accepting a proton from H2O.Is Acetate a base or acid?
Sodium acetate (CH3COONa) is a salt in solid state and can't be regarded as an acid or base in anhydrous or molten form. Although, being an ionic compound, sodium acetate dissociates in water to produce sodium ion Na+ and acetate ion CH3COO-.Is acetate positive or negative?
The acetate rod has lost electrons, giving it a positive charge. The duster has gained electrons, giving it a negative charge.What is acetate made of?
Acetate fabrics are made with spun filaments of cellulose taken from wood pulp. Classified as a chemical fiber textile or semi-synthetic, acetate is sometimes mixed with silk, wool or cotton to make it stronger. Acetate flakes are generated by a reaction of the wood pulp to a variety of acetic acids.What does acetate do in the body?
Release of acetate from tissues would occur under conditions when the function of the tricarboxylic acid cycle is restricted, so that the circulating acetate serves to redistribute oxidizable substrate throughout the body. This function is analogous to that served by ketone bodies.What is ch3choo?
CH3COO is the chemical formula for acetate. The products of these reactions are also commonly referred to as acetates such as sodium acetate. Acetate is the most common building block in biosynthesis, like the synthesis of fatty acids.Is acetate an element?
The acetate anion is formed from acetic acid and has a chemical formula of CH3COO-. The acetate anion is commonly abbreviated as OAc in formulas. For example, sodium acetate is abbreviated NaOAc and acetic acid is HOAc. The acetate ester group connects a functional group to the last oxygen atom of the acetate anion.How do you test for acetate?
Ferric Chloride[edit] HCl is added to the test tube till the precipitate has JUST dissolved. The resultant solution is added to the salt solution containing acetate to give a red colouration. The acetate ion is confirmed by adding water to this mixture and warming till formation of red precipitate.