What is hybridization of Mn in KMnO4?
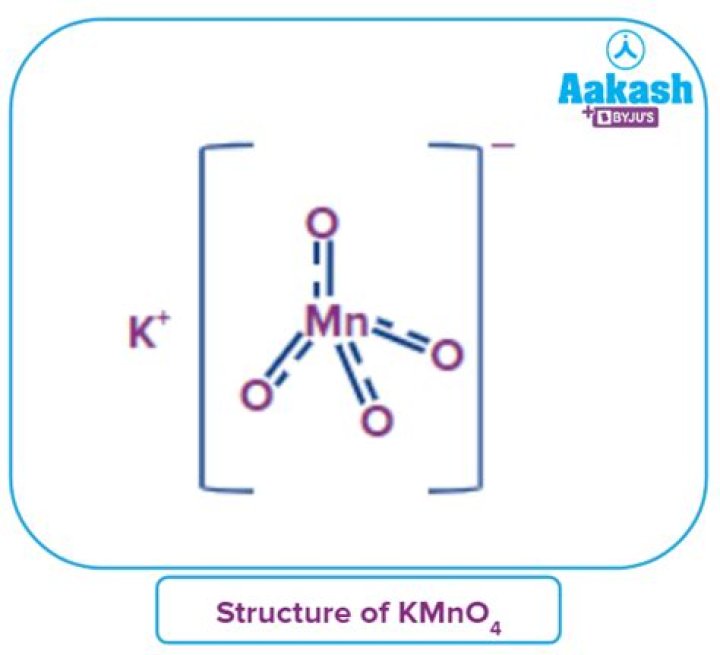
.
Consequently, what is the structure of MnO4?
Because manganese is in the +7 oxidation state, the permanganate(VII) ion is a strong oxidizing agent. The ion has tetrahedral geometry. Permanganate solutions are purple in color and are stable in neutral or slightly alkaline media.
One may also ask, why KMnO4 is Coloured? Potassium permanganate(KMnO4) is colored because it absorbs light in the visible range of the electromagnetic spectrum. This charge transfer takes place when a photon of light is absorbed, which leads to the purple color of the compound.
Also question is, how do you find the hybridization state of a compound?
Here's what you do: Look at the atom. Count the number of atoms connected to it (atoms – not bonds!) Count the number of lone pairs attached to it.
Add these two numbers together.
- If it's 4, your atom is sp3.
- If it's 3, your atom is sp2.
- If it's 2, your atom is sp.
What is the oxidation number of MnO4?
Manganate ions, or MnO4, have a charge of -1. Oxygen will usually have an oxidation number of -2. So the oxidation number of Mn in the MnO4 ion is +7.
Related Question AnswersWhy KMnO4 is a self indicator?
So once all the permanganate ions are used up in the reaction, the solution loses its pink colour. This indicates the end of the reaction and hence potassium permanganate is called a self indicator as it acts as an indicator apart from being one of the reactants.What Colour is mn2+?
The Mn2+ ions formed are almost colourless (very pale pink), so the end–point is the first permanent faint pink due to the first trace of excess of the brilliant purple manganate(VII) ion. Theoretically, there are actually two simultaneous colour changes, both masked by the redox indicator change.Why is it called permanganate?
, also known as manganate(VI) because it contains manganese in the +6 oxidation state. Manganates are the only known manganese(VI) compounds.Is MnO4 an acid or base?
Permanganate is a manganese oxoanion and a monovalent inorganic anion. It is a conjugate base of a permanganic acid. Generally purplish colored. Soluble in water.Why KMnO4 is added slowly in the titration?
This is because the concentration is quite large. When you start the titration, the dark purple KMnO is slowly added into the sample you are titrating. Under some heat and acidic conditions, this will cause the Mn which is in 7+ oxidation state in the permanganate to reduce into 2+ state as it reacts with the sample.What is the formula for Manganate?
Permanganate has the formula MnO4(-) i.e it has the central metal ion Mn in +VII oxidation state having electronic configuration [Ar] 3d(0)4s(0). While manganate anion has the formula of MnO4(2-). So here Mn has +VI oxidation state having one electron in the 3d energy level.What is permanganate used for?
Medical uses Potassium permanganate is used for a number of skin conditions. This includes fungal infections of the foot, impetigo, pemphigus, superficial wounds, dermatitis, and tropical ulcers.What's the charge of permanganate?
Re: Permanganate Ion Charge Here 1×(+7) and 4×(-2) hence the ion has -1. or 6eV for the first four electrons to four oxygen atoms.What do you mean by hybridisation?
In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals into new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory.How do you find the bond order?
If there are more than two atoms in the molecule, follow these steps to determine the bond order:- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
Does hybridization include lone pairs?
Lone pairs are electron groups which counts towards hybridization. Lone pairs count as one electron group towards total hybridization. Oxygen has two lone pairs. Along with the two bonded atoms, the hydrogen's, the central atom has a total of four electron groups, giving the central atom an sp3 hybridization.How do you determine the shape of a molecule?
Steps Used to Find the Shape of the Molecule- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons.
- Name the electron-group geometry.
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.



