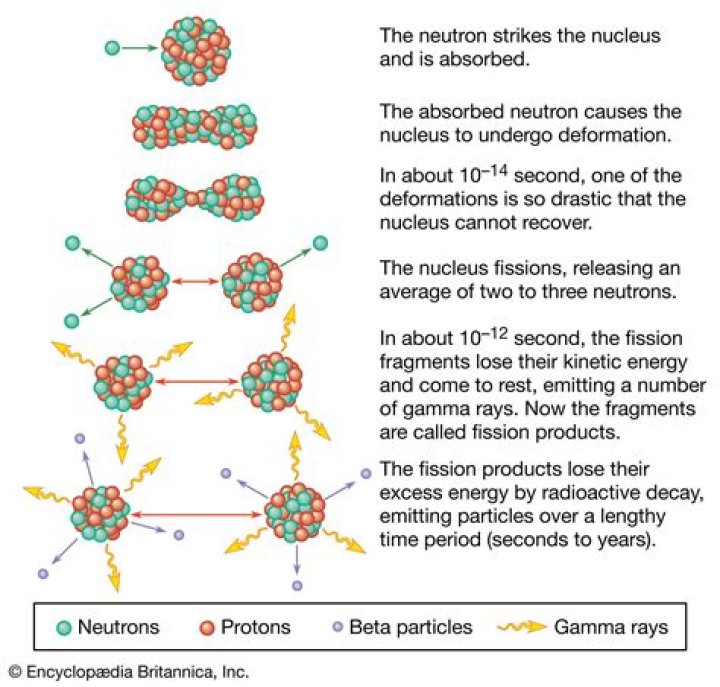
Nuclear fission is a nuclear reaction in which the nucleus of an atom splits into smaller parts (lighter nuclei). The fission process often produces free neutrons and photons (in the form of gamma rays), and releases a large amount of energy..
Also, what is nuclear fission reaction?
Nuclear fission is the process in which a large nucleus splits into two smaller nuclei with the release of energy. In other words, fission the process in which a nucleus is divided into two or more fragments, and neutrons and energy are released.
Furthermore, what is fission and example? Fission is the splitting of an atomic nucleus into two or more lighter nuclei accompanied by energy release. The energy released by nuclear fission is considerable. For example, the fission of one kilogram of uranium releases as much energy as burning around four billion kilograms of coal.
Likewise, what happens in a fission reaction?
Nuclear fission: In nuclear fission, an unstable atom splits into two or more smaller pieces that are more stable, and releases energy in the process. The fission process also releases extra neutrons, which can then split additional atoms, resulting in a chain reaction that releases a lot of energy.
What causes nuclear fission?
Nuclear fission either happens naturally or can be caused to happen by bombarding a fissionable isotope with neutrons. Nuclear fission happens when an atom splits into two atoms and releases energy.
Related Question Answers
What is fission used for?
Nuclear fission is a process whereby energy is released by the splitting of uranium atoms. Fission releases heat energy that can generate steam, which is used to spin a turbine to produce electricity. Nuclear Energy.How was Fission discovered?
What's not in doubt is that nuclear fission was first achieved by a team led by the German chemist Otto Hahn in 1938. Along with his colleague Fritz Strassmann, Hahn found that uranium atoms could be split by bombarding them with neutrons.Who discovered nucleus?
Ernest Rutherford
What is an example of fusion?
For example, uranium can fission to yield strontium and krypton. Fusion joins atomic nuclei together. The element formed has more neutrons or more protons than that of the starting material. For example, hydrogen and hydrogen can fuse to form helium.What is an example of nuclear fusion?
Nuclear fusion is when two or more lightweight atoms join together to form one heavier nucleus, with any energy released due to the conversion being converted into nuclear energy. An example of nuclear fusion is the process of four hydrogens coming together to form helium.What type of reaction is nuclear fusion?
Nuclear fusion is a reaction in which two or more atomic nuclei are combined to form one or more different atomic nuclei and subatomic particles (neutrons or protons). The difference in mass between the reactants and products is manifested as either the release or absorption of energy.How is fission controlled?
Controlled Nuclear Fission. To maintain a sustained controlled nuclear reaction, for every 2 or 3 neutrons released, only one must be allowed to strike another uranium nucleus. Most reactors are controlled by means of control rods that are made of a strongly neutron-absorbent material such as boron or cadmium.Why is fission dangerous?
These particles could then settle in the water tables, in food or clothing, or be inhaled in. The radioactive particles polluting the environment then decay, causing the radiation that is harmful to life. A building at the Fukushima nuclear fission power plant after an explosion.Does fission occur in the sun?
Fusion is what powers the sun. Both fission and fusion are nuclear reactions that produce energy, but the applications are not the same. Fission is the splitting of a heavy, unstable nucleus into two lighter nuclei, and fusion is the process where two light nuclei combine together releasing vast amounts of energy.What is K effective?
The multiplication factor that takes leakage into account is the effective multiplication factor (keff), which is defined as the ratio of the neutrons produced by fission in one generation to the number of neutrons lost through absorption and leakage in the preceding generation.Where does fission occur?
Radioactive fission, where the center of a heavy element spontaneously emits a charged particle as it breaks down into a smaller nucleus, does not occur often, and happens only with the heavier elements. Fission is different from the process of fusion, when two nuclei join together rather than split apart.Does nuclear fission happen naturally?
Natural nuclear fission occurs in really tiny amounts by the small amounts of natural U-235, some Thorium isotopes, Plutonium-239 and a few other “naturally fissile” materials. Uranium atoms ransomly fission all the time but they are naturally subcritical meaning that more neutrons are lost than produced.How does a nuclear reaction start?
By being bombarded with neutrons, nuclei with heavy atoms can be divided into several fragments formed by nuclei with lighter atoms, with neutron emission and a large release of energy. This type of nuclear reaction is called “nuclear fission reaction”.What does U 235 decay into?
Uranium-235 Decay Chain The decay chain of this radioactive metal is known as the Actinium Series withThorium-231 being the next isotope in this decay process. It makes Thorium-231 the daughter nuclide of this isotope. Uranium-235 is also known as Actinouranium as it is the parent isotope of the Actinium Series.What is a real life example of fission?
Fission is the process when an unstable and large element's nuclei disintegrate to form multiple smaller nuclei. A good example of a fission reaction is the nuclear power plant. In a nuclear power plant, this heat generated during fission is converted to electrical energy for our use in homes and factories.How is energy released in fission?
Nuclear fission is the process of splitting apart nuclei (usually large nuclei). When large nuclei, such as uranium-235, fissions, energy is released. So much energy is released that there is a measurable decrease in mass, from the mass-energy equivalence. This means that some of the mass is converted to energy.When was uranium discovered?
1789
Whats is an atom?
An atom a fundamental piece of matter. An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud.What is fission in science?
the act of cleaving or splitting into parts. Also called nuclear fission. Physics. the splitting of the nucleus of an atom into nuclei of lighter atoms, accompanied by the release of energy.