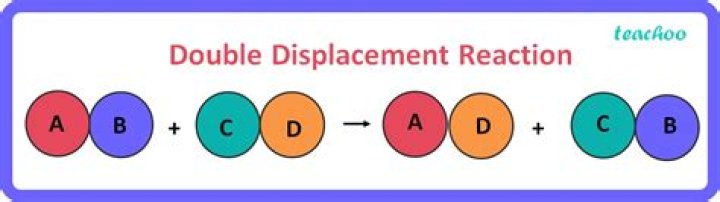
A double displacement reaction, also known as a double replacement reaction or metathesis, is a type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products..
In this regard, what is double replacement reaction in chemistry?
A double replacement reaction is a chemical reaction where two reactant ionic compounds exchange ions to form two new product compounds with the same ions.
what is double displacement reaction Class 10? Double Displacement Reaction:Reactions in which ions are exchanged between two reactants forming new compounds are called double displacement reactions. Double displacement reaction, in which precipitate is formed, is also known as precipitation reaction.
Also know, what is double displacement reaction with examples?
Double displacement reactions generally take place in aqueous solutions in which the ions precipitate and there is an exchange of ions. For example, on mixing a solution of barium chloride with sodium sulphate, a white precipitate of barium sulphate is immediately formed. These reactions are ionic in nature.
What is the other name of double displacement reaction?
metathesis reaction
Related Question Answers
What is an example of double replacement reaction?
A double-replacement reaction occurs when parts of two ionic compounds are exchanged, making two new compounds. A characteristic of a double-replacement equation is that there are two compounds as reactants and two different compounds as products. An example is. CuCl 2(aq) + 2 AgNO 3(aq) → Cu(NO 3) 2(aq) + 2 AgCl(s)What are the 3 types of double displacement reactions?
There are three types of reactions that fall under the double displacement reaction category: precipitation, neutralization and gas formation. A precipitation reaction forms an insoluble solid compound. A neutralization reaction occurs between an acid and a base that forms a salt and water.What are the examples of decomposition reaction?
A decomposition reaction occurs when one reactant breaks down into two or more products. This can be represented by the general equation: AB → A + B. Examples of decomposition reactions include the breakdown of hydrogen peroxide to water and oxygen, and the breakdown of water to hydrogen and oxygen.What is double displacement?
Double displacement Reaction - definition. A double displacement reaction is a type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products.What is the difference between displacement and double displacement reaction explain with example?
In a displacement reaction, a more reactive element replaces a less reactive element from a compound. In a double displacement reaction, two atoms or a group of atoms switch places form new compounds. In a displacement reaction, one displacement takes place. Salt solutions of two different metals react with each other.Why does double displacement occur?
A Double displacement(or double replacement) rxn or metathesis occurs bcz when two ionic compounds react and the positive ions(cation) and negative ions(anion) of the two reactants(must be ionic compounds) exchange their places, forming two new products(ionic compounds).Why double displacement reaction is called precipitation?
Because in most double displacement reaction of two ionic compounds lead to the formation of an insoluble product called precipitate. Hence it is known as precipitation reaction.What is displacement in chemistry?
Displacement reactions occur when a metal from the electrochemical series is mixed with the ions of a metal lower down in the electrochemical series. The atoms of the more reactive metal push their electrons on to ions of the less reactive metal. An example can be seen below.What are the products of a double displacement reaction?
In this reaction, A and C are positively-charged cations, while B and D are negatively-charged anions. Double-replacement reactions generally occur between substances in aqueous solution. In order for a reaction to occur, one of the products is usually a solid precipitate, a gas, or a molecular compound such as water.What is called displacement reaction?
Displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound. Both metals and non-metals take part in displacement reactions. Example : Reaction of iron nails with copper sulphate solution.How does double displacement work?
A double displacement reaction, also known as a double replacement reaction or metathesis reaction, is a type of reaction that occurs when the cations and anions switch between two reactants to form new products. A neutralization reaction occurs between an acid and a base that forms a salt and water.What are the types of displacement reaction?
A single-replacement reaction replaces one element for another in a compound. A double-replacement reaction exchanges the cations (or the anions) of two ionic compounds. A precipitation reaction is a double-replacement reaction in which one product is a solid precipitate.What is precipitation reaction?
A precipitation reaction is a type of chemical reaction in which two soluble salts in aqueous solution combine and one of the products is an insoluble salt called a precipitate. Also, a precipitate may form under certain conditions, but not others.Is Naoh HCL a double displacement reaction?
When sodium hydroxide (a base) reacts with hydrochloric acid, sodium chloride and water are formed. Double displacement reaction, in which precipitate is formed, is also known as precipitation reaction. Neutralisation reactions are also examples of double displacement reaction.