What is chlorination of methane?
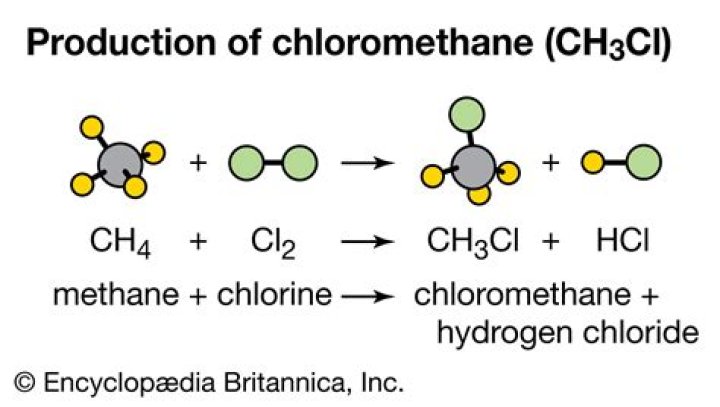
.
Simply so, what is chlorination reaction?
Illustrated Glossary of Organic Chemistry - Chlorination. Chlorination: Any reaction or process in which chlorine (and no other elements) are introduced into a molecule. Chlorination of an alkene by electrophilic addition of Cl2. Chlorination of a benzene ring by electrophilic aromatic substitution.
Subsequently, question is, why Ethane is produced in chlorination of methane? Chlorination of methane is a free radical reaction. During the process if two methyl radicals collapse they form ethane. So, ethane is also formed as a byproduct.
Additionally, what does Monochlorination mean?
monochlorination. Noun. (plural monochlorinations) (organic chemistry) chlorination with a single chlorine atom (where multiple chlorinations would be possible)
What are the products formed by the chlorination of methane in diffused sunlight?
Methane reacts with chlorine in diffused sunlight, or when heated to 600K to give chloromethane. Methyl chloride is formed by the substitution of hydrogen atom of a methane molecule, by a chlorine atom. This type of reaction is known as substitution reaction and the product formed is known as substitution product.
Related Question AnswersWhat are the types of chlorination?
The three most common types of chlorine used in water treatment are: chlorine gas, sodium hypochlorite, and calcium hypochlorite.What is the process of chlorination?
Chlorination is the process of adding chlorine to drinking water to disinfect it and kill germs. Different processes can be used to achieve safe levels of chlorine in drinking water. Chlorine is available as compressed elemental gas, sodium hypochlorite solution (NaOCl) or solid calcium hypochlorite (Ca(OCl)2 1.What is bromination reaction?
Bromination: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. Bromination of an alkene by electrophilic addition of Br2. Bromination of a benzene ring by electrophilic aromatic substitution. Bromination of a benzylic position by a free radical substitution reaction.What are 3 uses for chlorine?
Chlorine is commonly used as an antiseptic and is used to make drinking water safe and to treat swimming pools. Large amounts of chlorine are used in many industrial processes, such as in the production of paper products, plastics, dyes, textiles, medicines, antiseptics, insecticides, solvents and paints.What is halogenation reaction?
Halogenation is a reaction that occurs when one or more halogens are added to a substance. Halogens comprise the seventh column in the periodic table and include fluorine, chlorine, bromine, iodine, and astatine. The resulting product of a halogenation reaction is known as a halogenated compound.Why chlorine is added in water?
As a halogen, chlorine is a highly efficient disinfectant, and is added to public water supplies to kill disease-causing pathogens, such as bacteria, viruses, and protozoans, that commonly grow in water supply reservoirs, on the walls of water mains and in storage tanks.What is the mechanism of chlorination?
General mechanism The chain mechanism is as follows, using the chlorination of methane as a typical example: 1. Initiation: Splitting or homolysis of a chlorine molecule to form two chlorine atoms, initiated by ultraviolet radiation or sunlight. A chlorine atom has an unpaired electron and acts as a free radical.What is chlorine water formula?
Chlorine water is mainly just a solution of chlorine (Cl2) in water (H2O), with only a small amount of equilibrium disproportionation into HCl and HOCl..What is Monochlorination product?
Monochlorination of an alkane involves substituting one of the hydrogens in the alkane with a chlorine atom. Theses conditions cause the chlorine molecule to split into chlorine free radicals. Monochlorination is typically, not hydrogen selective like other reactions in orgo.How many Monochlorination products are possible?
Here, we have 5 constitutional isomers possible (not counting stereoisomers). Again, it helps to break out your IUPAC nomenclature to double-check that there are no duplicates. It is impossible to capture the variety of potential questions with these three examples, but the general thrust is the same.What is a Monohalogenated alkane?
O A Monohalogenated Alkane Is A Haloalkane Containing Only One Halogen Atom. A Monohalogenated Alkane Is A Haloalkane Containing Several Identical Halogen Atoms.What are Monochloro derivatives?
Monochloro derivatives means C-12 sharing at least and at most one chlorine bond. In an alkane every C atom can have at least and at most one chlorine bond. So in n-pentane the will be 5 monochloro derivatives.How many alkyl chlorides can be obtained from Monochlorination?
64 Cards in this Set| show the initiation, propagation, and termination steps for the monochlorination of cyclohexane. | 11.1 | |
|---|---|---|
| . | . | 11.4 |
| How many alkyl chlorides can be obtained from monochlorination of the following alkanes? Disregard stereoisomers. | 11.5 |



