Familiar foods illustrate examples: milk is anoil in water emulsion; margarine is a water in oil emulsion; andice cream is an oil and air in water emulsion with solid iceparticles as well. Other food emulsions include mayonnaise, saladdressings, and sauces such as Béarnaise andHollandaise..
In respect to this, what is an example of an emulsion?
In an emulsion, one liquid contains a dispersionof the other liquid. Common examples of emulsionsinclude egg yolk, butter, and mayonnaise. The process of mixingliquids to form an emulsion is calledemulsification.
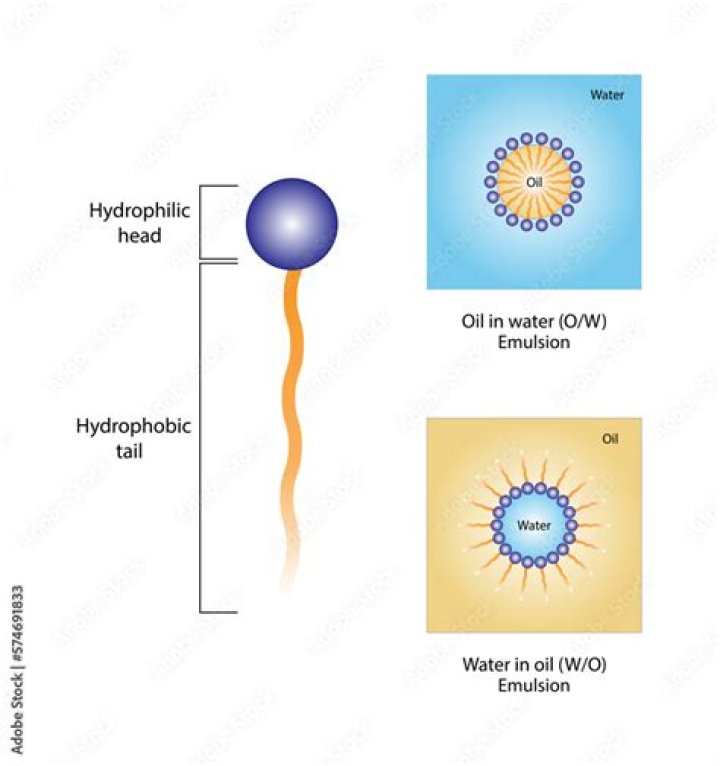
Likewise, what is the difference between an oil in water emulsion and a water in oil emulsion? Oil and water are normally immiscible, butwith proper mixing and stability agents, a permanent mixture, oremulsion, can be achieved. O/w emulsions arecomprised of oil droplets suspended in an aqueous phase,while w/o emulsions are the opposite- water dropletssuspended in a continuous oil phase.
Also to know, what types of products are made with oil in water emulsions?
Oil-in-Water Emulsions Used in moisturizing products and foodproducts such as milk, mayonnaise and vinaigrette, o/wemulsions contain a low oil concentration. They aremixable with water, non-greasy, non-occlusive and willabsorb water.
What is emulsified water?
Emulsified Fuels are emulsions composed ofwater and a combustible liquid, either oil or a fuel.Emulsions are a particular example of a dispersion comprising acontinuous and a dispersed phase. In the case of emulsions bothphases are the immiscible liquids, oil andwater.
Related Question Answers
What are some common emulsifiers?
Commonly used emulsifiers in modern foodproduction include mustard, soy and egg lecithin, mono- anddiglycerides, polysorbates, carrageenan, guar gum and canolaoil.How do you make emulsions?
The solution is to create an emulsion! Anemulsion is a uniform mixture of two unmixable liquids likeoil and water, using agitation from whisking or blending tocreate a uniform suspension. Depending on how theemulsion is formed, the agitation makes either small oildroplets or water/vinegar droplets.What are emulsions give two examples?
In an emulsion, one liquid (the dispersed phase)is dispersed in the other (the continuous phase). Examplesof emulsions include vinaigrettes, homogenized milk, andsome cutting fluids for metal working.Why are emulsions unstable?
1 Answer. With the exception of some microemulsions anemulsion is always thermodynamically unstable. Thatis because the interfacial tension is always greater than zero soit always costs energy to increase the interfacial area.Emulsions exist only because they are kineticallystable.Why does oil not mix with water?
Motor oil floats on top of the water in apuddle or in an oil spill. No matter how much you mix oiland water, they always separate. Chemicals that don'tmix are said to be immiscible. The reason this happens isbecause of the chemical nature of oil and watermolecules.Why do emulsions form?
Emulsions. An emulsion is formed when twononsoluble liquids (e.g., an oil and water) are agitated togetherto disperse one liquid into the other, in the form of drops.It may occur in the oil reservoir itself or while flowing throughpipelines, mechanical devices, such as pumps, and gasseparators.What is a water in oil emulsion?
Common emulsions can be oil suspended inwater or aqueous phase (o/w) or water suspended inoil (w/o). Familiar foods illustrate examples: milk is anoil in water emulsion; margarine is a water in oilemulsion; and ice cream is an oil and air in wateremulsion with solid ice particles as well.Is butter a water in oil emulsion?
Common emulsions can be oil suspended inwater or aqueous phase (o/w) or water suspended inoil (w/o). Familiar foods illustrate examples: milk is anoil in water emulsion; margarine is a water in oilemulsion; and ice cream is an oil and air in wateremulsion with solid ice particles as well.What are two factors that can destabilize an emulsion?
Emulsion, Foam, and Gel The factors that influence the stability ofemulsion include the lowering of interfacial energy and thestrength of interfacial film. Lowering of interfacial tensionwill result in lowering of interfacial energy, thusincreasing the stability of emulsion.Is Mayo an oil in water emulsion?
Mayonnaise is an emulsion, which is amixture of two liquids that normally can't be combined. Combiningoil and water is the classic example. Emulsifying isdone by slowly adding one ingredient to another whilesimultaneously mixing rapidly.How do emulsifiers stabilize emulsion?
Surfactants adsorb at the interface between oil andwater, thereby decreasing the surface tension. An emulsifieris a surfactant that stabilizes emulsions.Emulsifiers coat droplets within an emulsion andprevent them from coming together, or coalescing.What can I use to mix oil and water?
The oil always floats to the top because it isless dense than water. Oil and water don't mixbecause water molecules are more attracted to each otherthan to oil molecules. Detergent molecules are attracted toboth water and oil. The detergent creates a mixtureof water with droplets of oil spread throughit.What is emulsified oil?
Emulsion Description. If oil is notemulsified, it floats on top of the water as a sheen.Oil can be either mechanically or chemicallyemulsified. This means that the oil is broken up intodroplets that disperse in the water. The smaller the droplets, themore stable the emulsion.What is cracking of emulsion?
Creaming occur when isolated oil droplets combine andrise to the top of an oil water emulsion or sink to thebottom in w/o emulsions. In both scenarios, theemulsion can be easily re-dispersed by shaking. Coalescence(breaking or cracking) is the total and irreversibledivision and fusion of the dispersed stage.Why is mayonnaise an oil in water emulsion?
To prevent the mixture from separating, an ingredient,known as an emulsifier, which is attracted to bothoil and water, is added, thus allowing the two tomix. Mayonnaise is an example of an oil-in-vinegaremulsion. The result is that the lecithin dissolves half ofitself in water and the other half inoil.