updates /
What is alkyl alkanoate?
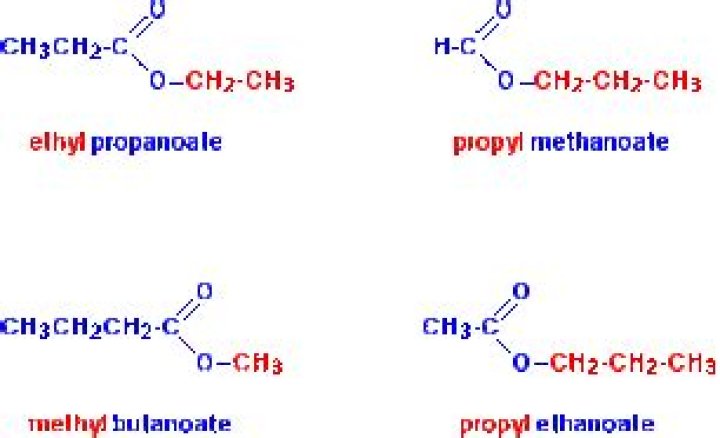
esters. alkyl alkanoate (a two-part name) alkyl group is attached to the O. alkanoate analogous to carboxylate salt.
.
Considering this, what is Alkanoate?
alkanoate. Noun. (plural alkanoates) (organic chemistry) Any salt or ester of an alkanoic acid.
Furthermore, how do you name Alkanoate? Naming Alkanoates Esters are generally derived from a carboxylic acid and an alcohol. Carboxylic acids = COOH, in esters the hydrogen is replaced by a hydrocarbon (eg Ethyl Ethanoate.) Note: the acid is named by counting up the total number of carbon atoms in the chain - including the one in the -COOH group.
Accordingly, what is Alkanoate in chemistry?
Noun. alkanoate (plural alkanoates) (organic chemistry) Any salt or ester formed from the reaction of an alkanoic acid and alkanol.
What is the functional group of Alkanoate?
Groups containing oxygen
| Chemical class | Group | Suffix |
|---|---|---|
| Carboxylate | Carboxylate | -oate |
| Carboxylic acid | Carboxyl | -oic acid |
| Ester | Carboalkoxy | alkyl alkanoate |
| Methoxy | Methoxy |
What do you mean by esterification?
Esterification is a chemical reaction that forms at least one ester (= a type of compound produced by reaction between acids and alcohols). Esters are produced when acids are heated with alcohols in a process called esterification. An ester can be made by an esterification reaction of a carboxylic acid and an alcohol.Why does ethyl Ethanoate have a low boiling point?
Ester molecules are polar but have no hydrogen atom attached directly to an oxygen atom. They are therefore incapable of engaging in intermolecular hydrogen bonding with one another and thus have considerably lower boiling points than their isomeric carboxylic acids counterparts. Esters are common solvents.What is the formula of Ester?
Esters have the general formula RCOOR′, where R may be a hydrogen atom, an alkyl group, or an aryl group, and R′ may be an alkyl group or an aryl group but not a hydrogen atom. (If it were hydrogen atom, the compound would be a carboxylic acid.)How do we name Esters?
Esters can be named using a few steps Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain from the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.What are the properties of esters?
Physical properties and characterization Esters are more polar than ethers but less polar than alcohols. They participate in hydrogen bonds as hydrogen-bond acceptors, but cannot act as hydrogen-bond donors, unlike their parent alcohols. This ability to participate in hydrogen bonding confers some water-solubility.How do you name carboxylic acids?
In general, carboxylic acids are named based on the number of carbons in the longest continuous chain, including the carboxyl group (-COOH). The suffix of this carbon chain is then replaced, as carboxylic acids always end in "-oic acid." An example is CH2O2, in which the longest continuous carbon chain is a methane.How do you name anhydrides?
Unsymmetrical acid anhydrides are named by first naming each component carboxylic acid alphabetically arranged (without the word acid) followed by spaces and then the word anhydride.How do you make esters?
Esters are thus usually prepared by the reaction of carboxylic acids, or their derivatives, and alcohols. One of the simplest ways to get an ester is the boiling of a carboxylic acid dissolved in an alcohol with a catalytic amount of a mineral acid.How do you name amide?
Primary amides are named by changing the name of the acid by dropping the -oic acid or -ic acid endings and adding -amide. The carbonyl carbon is given the #1 location number.Is Ester a functional group?
Esters are a functional group commonly encountered in organic chemistry. They are characterized by a carbon bound to three other atoms: a single bond to a carbon, a double bond to an oxygen, and a single bond to an oxygen. Esters react with nucleophiles at the carbonyl carbon.What are the 7 functional groups?
There are 7 important functional groups in the chemistry of life: Hydroxyl, Carbonyl, Carboxyl, Amino, Thiol, Phosphate, and aldehyde groups.- Hydroxyl group: consists of a hydrogen atom covalently bonded to an oxygen atom.
- Carbonyl group: is written as a covalent C=O.



