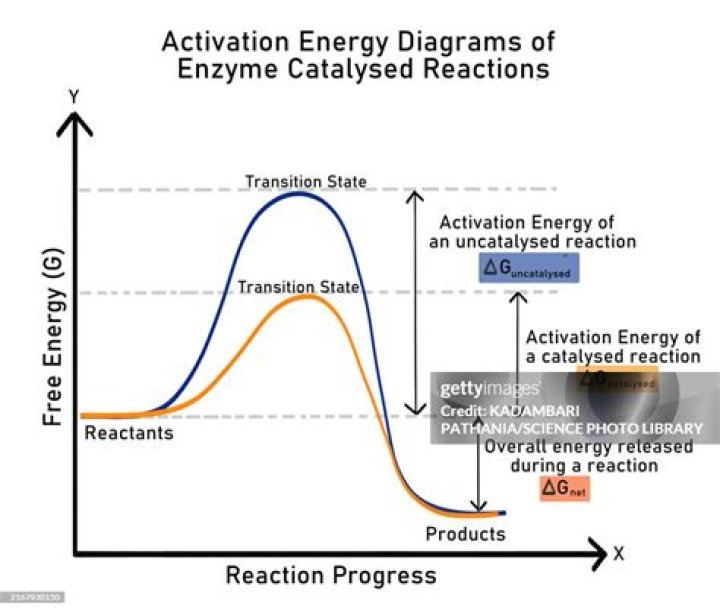
Enzymes are biological catalysts. Catalysts lower the activation energy for reactions. The lower the activation energy for a reaction, the faster the rate. Thus enzymes speed up reactions by lowering activation energy..
Accordingly, what is activation energy enzymes?
The activation energy is the energy required to start a reaction. Enzymes are proteins that bind to a molecule, or substrate, to modify it and lower the energy required to make it react. The rate of reaction is given by the Arrhenius equation.
Similarly, how does an enzyme affect the energy of a chemical reaction? Explanation: Enzymes are catalysts which will lower the activation energy of a chemical reaction. Activation energy is the energy needed to get a reaction going. By lowering the amount of energy needed to start a reaction, the reaction can go more quickly.
Keeping this in consideration, how do enzymes lower the activation energy of a reaction?
The activation energy is the amount of energy needed for the reaction to go forward and get over its activation barrier. Enzymes lower the activation energy of a reaction by binding one of the reactants, called a substrate, and holding it in a way that lowers the activation energy.
How does enzyme work?
Enzymes are biological molecules (typically proteins) that significantly speed up the rate of virtually all of the chemical reactions that take place within cells. The molecules that an enzyme works with are called substrates. The substrates bind to a region on the enzyme called the active site.
Related Question Answers
Why is activation energy important?
All chemical reactions, including exothermic reactions, need activation energy to get started. Activation energy is needed so reactants can move together, overcome forces of repulsion, and start breaking bonds.What is the best definition of activation energy?
Which is the best definition of activation energy? the energy required to end a chemical reaction. the energy required to bind a substrate to an active site. the energy required to break the bonds of reactant molecules.How do you find the activation energy?
The value of the slope (m) is equal to -Ea/R where R is a constant equal to 8.314 J/mol-K. The activation energy can also be found algebraically by substituting two rate constants (k1, k2) and the two corresponding reaction temperatures (T1, T2) into the Arrhenius Equation (2).What factors affect activation energy?
Reactions occur when two reactant molecules effectively collide, each having minimum energy and correct orientation. Reactant concentration, the physical state of the reactants, and surface area, temperature, and the presence of a catalyst are the four main factors that affect reaction rate.How do you draw an activation energy diagram?
1 Answer - Draw and label a pair of axes. Label the vertical axis "Potential Energy" and the horizontal axis "Reaction Coordinate".
- Draw and label two short horizontal lines to mark the energies of the reactants and products.
- Draw the energy level diagram.
- Draw and label the activation energy.
What is an example of the use of activation energy?
A Common Example You have probably used activation energy to start a chemical reaction. For example, if you've ever struck a match to light it, then you provided the activation energy needed to start a combustion reaction. When you struck the match on the box, the friction started the match head burning.What affects enzyme activity?
Several factors affect the rate at which enzymatic reactions proceed - temperature, pH, enzyme concentration, substrate concentration, and the presence of any inhibitors or activators.What is the role of activation energy?
Activation energy is defined as the minimum amount of energy required by the reactant molecules to undergo a chemical reaction. But when we add a catalyst into the reaction then there will be a decrease in activation energy and as a result, molecules will lesser amount of energy are able to participate in the reaction.How does a catalyst lower the activation energy?
The function of a catalyst is to lower the activation energy so that a greater proportion of the particles have enough energy to react. A catalyst can lower the activation energy for a reaction by: reacting with the reactants to form an intermediate that requires lower energy to form the product.What is the definition of activation energy in chemistry?
The term Activation Energy was introduced in 1889 by Svante Arrhenius, a Swedish scientist. It is defined as the least possible amount of energy (minimum) which is required to start a reaction or the amount of energy available in a chemical system for a reaction to take place.How does a catalyst affect activation energy?
Catalysts provide a new reaction pathway in which a lower A.E. is offered. A catalyst increases the rate of a reaction by lowering the activation energy so that more reactant molecules collide with enough energy to surmount the smaller energy barrier.What do you mean by enzymes?
Enzyme: Proteins that speeds up the rate of a chemical reaction in a living organism. An enzyme acts as catalyst for specific chemical reactions, converting a specific set of reactants (called substrates) into specific products. Without enzymes, life as we know it would not exist.How are enzymes produced?
Enzymes are made from amino acids, and they are proteins. When an enzyme is formed, it is made by stringing together between 100 and 1,000 amino acids in a very specific and unique order. The chain of amino acids then folds into a unique shape. Other types of enzymes can put atoms and molecules together.What is an example of an enzyme?
An enzyme's name is often derived from its substrate or the chemical reaction it catalyzes, with the word ending in -ase. Examples are lactase, alcohol dehydrogenase and DNA polymerase. Different enzymes that catalyze the same chemical reaction are called isozymes.How does pH affect enzyme activity?
pH: Each enzyme has an optimum pH range. Changing the pH outside of this range will slow enzyme activity. Extreme pH values can cause enzymes to denature. Enzyme concentration: Increasing enzyme concentration will speed up the reaction, as long as there is substrate available to bind to.How do enzymes catalyze reactions?
Enzyme catalysisAn enzyme catalyzes a biochemical reaction by binding a substrate at the active site. After the reaction has proceeded, the products are released and the enzyme can catalyze further reactions.What do enzymes increase?
Enzymes are biological catalysts. Catalysts lower the activation energy for reactions. The lower the activation energy for a reaction, the faster the rate. Thus enzymes speed up reactions by lowering activation energy.What are all the chemical reactions in the body?
Answer and Explanation: The sum of all chemical reactions in the body is known as metabolism. It can be divided into two branches namely catabolism and anabolism.What do you mean by free energy?
In physics and physical chemistry, free energy refers to the amount of internal energy of a thermodynamic system that is available to perform work. Helmholtz free energy is energy that may be converted into work at constant temperature and volume.