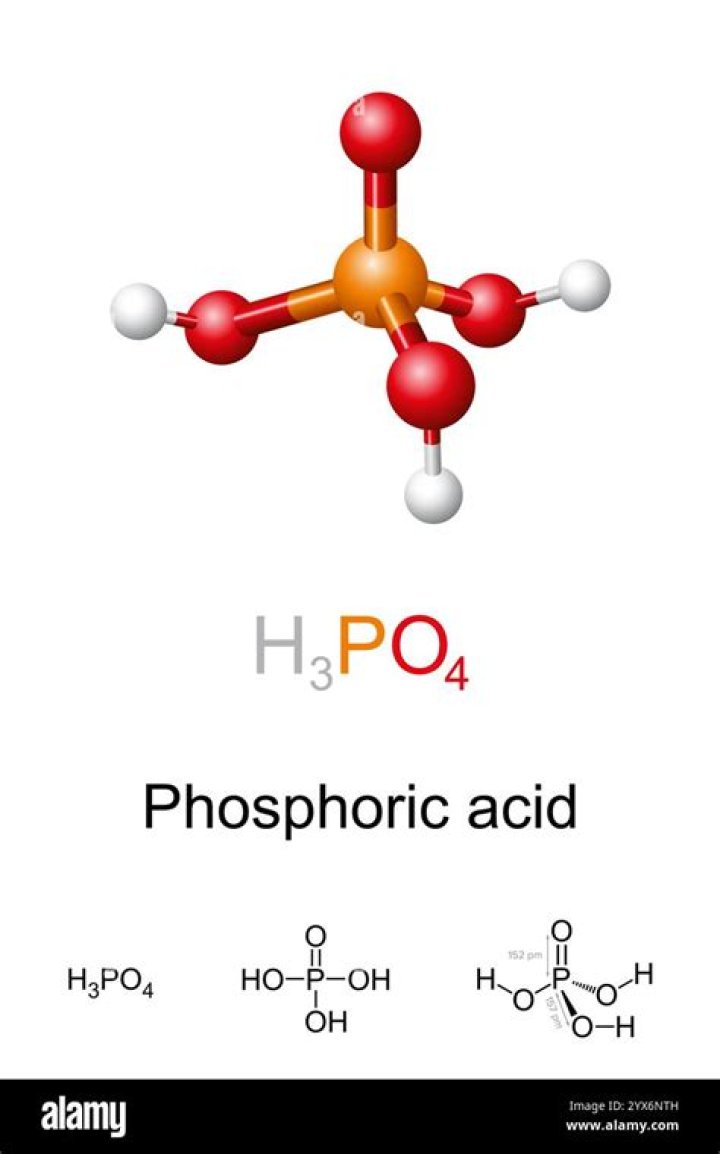
Phosphoric acid has basicity of 3 i.e it canloose 3 H+ while phosphorous acid has basicity of 2.Phosphoric acid liberates more protons than phosphorousacid (as the basicity of phosphoric acid is 3 andthat of phosphorous acid is 2)..
Accordingly, is h3po4 a strong or weak acid?
Phosphorus is not electronegative enough. Along with thehigher electronegativity of N relative to P, the extra oxygen makesHNO3 a strong acid, while the lack thereof makes H3PO4weak. After H3PO4 does lose a proton, it forms H2PO4-,which lacks sufficient resonance stabilization.
Beside above, why is h3po3 more acidic than h3po4? H3PO3 is more polar, so the O-H bonds willbe weaker, making the H+ easier to come off, and causingH3PO3 to be more acidic than H3PO4.
Also to know, which is more acidic h3po4 or H3AsO4?
Comparing Acidic Strength of H3PO4 andH3AsO4. The pKa values for H3AsO4 and H3PO4are 2.30 and 2.12 which indicate that H3PO4 is a strongeracid compared to H3AsO4.
What is phosphoric acid pH?
Phosphoric Acid is a colorless, odorlessphosphorus-containing inorganic acid. It is a conjugateacid of a dihydrogenphosphate and a phosphate ion. fromChEBI. Phosphoric acid appears as a clear colorless liquidor transparent crystalline solid. The pure solid melts at42.35°C and has a density of 1.834 g / cm3.
Related Question Answers
What are the 7 strong acids?
Terms in this set (7) - hydrochloric acid. HCL.
- hydrobromic acid. HBr.
- Hydroiodic acid. HI.
- Nitric acid. HNO3.
- Perchloric acid. HClO4.
- Sulfuric acid. H2SO4.
- Chloric acid. HClO3.
What is the world's strongest acid?
The carborane superacids may be considered theworld's strongest solo acid, as fluoroantimonicacid is actually a mixture of hydrofluoric acid andantimony pentafluoride. Carborane has a pH value of-18.Is h3po4 acidic or basic?
Acidic properties Phosphoric acid, H3PO4, isa tribasic acid.What are the six strong acids?
Terms in this set (6) - H2SO4. Sulfuric Acid.
- HCl. Hydrochloric Acid.
- HBr. Hydrobromic Acid.
- HI. Hydroiodic Acid.
- HNO3. Nitric Acid.
- HClO4. Perchloric Acid.
Is HClO4 strong or weak?
If you see any other acid or base than one of thesestrong ones it will be a weak acid or base (unless Ispecifically say otherwise in the problem). The 7 commonstrong acids are: HCl, HBr, HI, HNO3, HClO3, HClO4and H2SO4 (1st proton only).Is LiOH strong or weak?
Classifying Electrolytes
| Strong Electrolytes | strong acids | HCl, HBr, HI, HNO3, HClO3,HClO4, and H2SO4 |
| strong bases | NaOH, KOH, LiOH, Ba(OH)2, andCa(OH)2 |
| salts | NaCl, KBr, MgCl2, and many, many more |
| Weak Electrolytes |
| weak acids | HF, HC2H3O2 (acetic acid),H2CO3 (carbonic acid),H3PO4 (phosphoric acid), and many more |
What are the strong bases?
Here is a list of the most common strong bases. - LiOH - lithium hydroxide.
- NaOH - sodium hydroxide.
- KOH - potassium hydroxide.
- RbOH - rubidium hydroxide.
- CsOH - cesium hydroxide.
- *Ca(OH)2 - calcium hydroxide.
- *Sr(OH)2 - strontium hydroxide.
- *Ba(OH)2 - barium hydroxide.
Is NaOH a strong or weak base?
Sodium hydroxide (NaOH) is strong basebecause it fully dissociates in water to produce hydroxide ions.While weak bases produce fewer hydroxide ions, making thesolution less basic.Is h2so4 stronger than h3po4?
This is due to the fact that H2SO4dissociates/deprotonates more in H2O than H3PO4 does.Stronger acids deprotonate almost completely in water, whereweak acids dissociate incompletely in water, making H2SO4 astronger acid, as the first Hydrogen atom dissociatescompletely, and the second does partially.Which is more acidic h2so4 or H2SeO4?
oxyacids of sulphur is more acidic then seleniumbecause sulphur is more electronegative then selenium.HenceH2SO4 should be stronger than H2SeO4.Which acid is stronger h2s or H2Te?
Therefore, acidic character increases. H2Te hasless bond dissociation enthalpy than H2S . So, less energyis required to break H2Te bond & releasing [H]+ iseasier & hence the acidity of H2Te is higher. Whereas,H2S has High Bond Dissociation Energy & hence theacidity of H2S is less.Is H3AsO4 a strong acid?
Nominally a stronger acid is one that releasesfree proton more readily. H3AsO4 (arsenic acid) is amuch stronger acid than H3AsO3 (arsenous acid). Thefirst pKa for arsenic acid is 2.2 while for arsenous it is9.2. 2.2 is a moderately strong acid. 9.2 is very weakacid.Is arsenic acid strong or weak?
Acids are considered weak if they have apKa value greater than -2, or pH greater than 2 but less than 7.Arsenic acid, boric acid, linolenic acid,phosphoric acid, selenious acid, hydrofluoricacid.What is the name of H3AsO3?
Substance Name: Arsenious acid (H3AsO3),trisodium salt (8CI)Why is phosphorous acid stronger than phosphoric acid?
In phosphorus acid, there is a H on the maincarbon. Hydrogen doesn't pull as much on electrons as Oxygens doand hence that part of the molecule becomes more positive causing astronger dipole moment than that in phosphoricacid.What is the basicity of h3po4?
Basicity of an acid refers to the number ofreplaceable hydrogen atoms in one molecule of the acid. As you cansee in the above image of structure of H3PO4, there are 3OH's attached. Thus the basicity of H3PO4 is also3.Is phosphoric acid more acidic than citric acid?
For the first proton, phosphoric acid isstronger (Ka1 = 7.5×10^-3 for phosphoric acidvs. 7.4×10^-4 for citric acid). However, the secondand third protons are both stronger in citric acid(Ka2 = 1.7×10^-5 for citric vs. 6.2×10^-8 forphosphoric and Ka3 = 4.0×10^-7 for citric acidvs. 4.2×10^-13 for phosphoric acid).What is the pKa of phosphoric acid?
The three pKa values for phosphoric acid(from the CRC Handbook of Chemistry and Physics) are 2.16, 7.21,and 12.32.Is h3po2 a strong acid?
Originally Answered: What is the acidic strengthorder of h3po2, h3po3, and h3po4? In some textbooks, generalrule followed is higher the oxidation state of central atom, moreis the acidic nature.