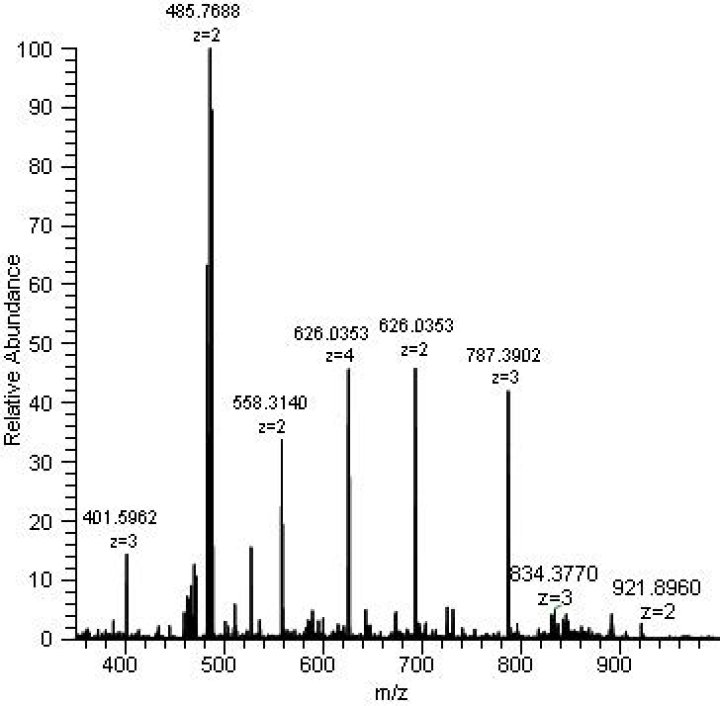
A mass spectrum is an intensity vs. m/z (mass-to-charge ratio) plot representing a chemical analysis. Hence, the mass spectrum of a sample is a pattern representing the distribution of ions by mass (more correctly: mass-to-charge ratio) in a sample..
Consequently, what is mass spectrometry in chemistry?
Mass spectrometry (MS) is an analytical technique that measures the mass-to-charge ratio of ions. These spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical identity or structure of molecules and other chemical compounds.
Likewise, what are the peaks in a mass spectrum? A mass spectrum will usually be presented as a vertical bar graph, in which each bar represents an ion having a specific mass-to-charge ratio (m/z) and the length of the bar indicates the relative abundance of the ion. The most intense ion is assigned an abundance of 100, and it is referred to as the base peak.
Also Know, how does a mass spectrum work?
A mass spectrometer produces charged particles (ions) from the chemical substances that are to be analyzed. The mass spectrometer then uses electric and magnetic fields to measure the mass ("weight") of the charged particles.
What is the application of mass spectrometry?
Mass spectrometry represents a powerful technique with a myriad of different applications in biology, chemistry, and physics, but also in clinical medicine and even space exploration. It is used to determine the molecular weight of compounds by separating molecular ions on the basis of their mass and charge.
Related Question Answers
Where is mass spectrometry used?
Specific applications of mass spectrometry include drug testing and discovery, food contamination detection, pesticide residue analysis, isotope ratio determination, protein identification, and carbon dating.Why is mass spectrometry important?
Mass spectrometry is playing a significant role in organometallic chemistry. One of the most important applications of mass spectrometry is the determination of the molecular mass and elemental composition of metal compounds, and the identification of their structure.What is the difference between mass spectrometry and mass spectroscopy?
Mass spectrometry (MS) is an analytical technique that ionizes chemical species and sorts the ions based on their mass-to-charge ratio. In simpler terms, a mass spectrum measures the masses within a sample. Mass spectrometry is used in many different fields and is applied to pure samples as well as complex mixtures.What are the different types of mass spectrometry?
Types of Mass Spectrometry[edit] - AMS (Accelerator Mass Spectrometry)[edit]
- Gas Chromatography-MS[edit]
- Liquid Chromatography-MS[edit]
- ICP-MS (Inductively Coupled Plasma-Mass spectrometry )[edit]
- IRMS (Isotope Ratio Mass Spectrometry)[edit]
- Ion Mobility Spectrometry-MS[edit]
- MALDI-TOF[edit]
- SELDI-TOF[edit]
How do you find the mass to charge ratio?
In mass spectroscopy, the mass-to-charge ratio (symbols: m/z, m/e) of a cation is equal to the mass of the cation divided by its charge. Since the charge of cation formed in the mass spectrometer is almost always +1, the mass-to-charge ratio of a cation is usually equal to the mass of the cation.How abundance is measured in a mass spectrometer?
Isotopes have different atomic masses. The relative abundance of each isotope can be determined using mass spectrometry. A mass spectrometer ionizes atoms and molecules with a high-energy electron beam and then deflects the ions through a magnetic field based on their mass-to-charge ratios ( m / z m/z m/z ).Is mass spectrometry quantitative or qualitative?
Mass spectrometry is not inherently quantitative because of differences in the ionization efficiency and/or detectability of the many peptides in a given sample, which has sparked the development of methods to determine relative and absolute abundance of proteins in samples.How do you calculate Monoisotopic mass?
The theoretical monoisotopic mass of a molecule is computed by taking the sum of the accurate masses of the primary isotope of each atom in the molecule. For small molecules made up of low atomic number elements the monoisotopic mass is observable as an isotopically pure peak in a mass spectrum.What is the M+ peak in mass spectrometry?
M+=28 Most atoms except N have either: a) odd valency and odd mass (H, B, P, halides) or (b) even valency and even mass (C, O, S, Si). The M+ peak is usually the highest intensity peak in the cluster of peaks at highest m/z.What is meant by McLafferty rearrangement?
The McLafferty rearrangement is an organic reaction seen in mass spectrometry. The McLafferty rearrangement is an example of a hydrogen atom jumping to the other fragment as a part of the process of the bond breaking. It happens in an organic molecule containing a keto-group.What is the mass spectrum of chlorine?
The mass spectrum of chlorine Chlorine has two isotopes, 35Cl and 37Cl, in the approximate ratio of 3 atoms of 35Cl to 1 atom of 37Cl. You might suppose that the mass spectrum would look like this: You would be wrong! The problem is that chlorine consists of molecules, not individual atoms.Why does fragmentation occur in mass spectrometry?
Fragmentation (mass spectrometry) In mass spectrometry, fragmentation is the dissociation of energetically unstable molecular ions formed from passing the molecules in the ionization chamber of a mass spectrometer. The fragments of a molecule cause a unique pattern in the mass spectrum.What does a mass spectrum show?
A mass spectrum is simply the m/z ratios of the ions present in a sample plotted against their intensities. Each peak in a mass spectrum shows a component of unique m/z in the sample, and heights of the peaks connote the relative abundance of the various components in the sample.How is mass spectrometry used in drug testing?
The instrument is an analytical tool known as a mass spectrometer. It tests the urine for the presence of the steroid or chemicals that are produced as the body processes the steroid, which may be detected for days to weeks after the last dose, depending on the drug and the amount taken.What is the difference between atomic mass and atomic weight?
Atomic mass (ma) is the mass of an atom. A single atom has a set number of protons and neutrons, so the mass is unequivocal (won't change) and is the sum of the number of protons and neutrons in the atom. Atomic weight is a weighted average of the mass of all the atoms of an element, based on the abundance of isotopes.What does a mass spectrum tell you?
A mass spectrum is a plot of the ion signal as a function of the mass-to-charge ratio. These spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical identity or structure of molecules and other chemical compounds.What is the basic principle of mass spectrometry?
Basic Principle A mass spectrometer generates multiple ions from the sample under investigation, it then separates them according to their specific mass-to-charge ratio (m/z), and then records the relative abundance of each ion type.What is nitrogen rule in mass spectrometry?
The nitrogen rule states that organic compounds containing exclusively hydrogen, carbon, nitrogen, oxygen, silicon, phosphorus, sulfur, and the halogens either have 1) an odd nominal mass that indicates an odd number of nitrogen atoms are present or 2) an even nominal mass that indicates an even number of nitrogenWhat does M+ mean in mass spec?
mass spectra - the molecular ion (M+) peak. MASS SPECTRA - THE MOLECULAR ION (M+) PEAK. This page explains how to find the relative formula mass (relative molecular mass) of an organic compound from its mass spectrum. It also shows how high resolution mass spectra can be used to find the molecular formula for a