What is a hydrogen bond acceptor?
.
In this manner, what makes a hydrogen bond acceptor?
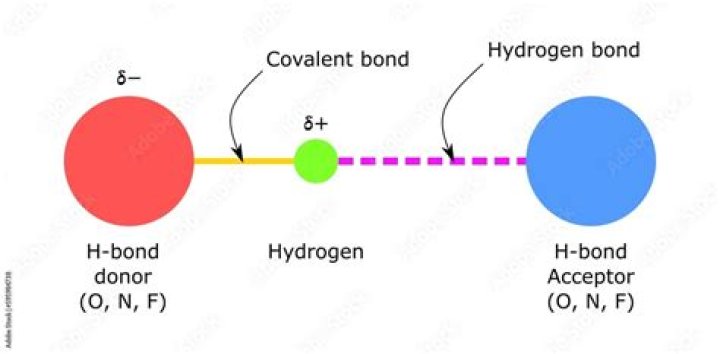
Hydrogen bonds are formed between a hydrogen atom bound to a small, highly electronegative atom and another small, highly electronegative atom with an unshared electron pair. The nitrogen atom is called the hydrogen bond acceptor, because it is "accepting" the hydrogen from the oxygen.
Secondly, what is a hydrogen bond between? A hydrogen bond (often informally abbreviated H -bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative atom or group, particularly the second-row elements nitrogen (N), oxygen (O), or fluorine (F)—the hydrogen bond donor (Dn)—and another
Keeping this in view, is an ether a hydrogen bond acceptor?
Ether is a hydrogen bond acceptor. Two ether molecules cannot make hydrogen bonds as both of them do not have hydrogen attached to oxygen but they can make bonds with other molecules capable of hydrogen bonding such as alcohols, water, etc.
What is an example of hydrogen bond?
A ubiquitous example of a hydrogen bond is found between water molecules. In a discrete water molecule, there are two hydrogen atoms and one oxygen atom. Water is unique because its oxygen atom has two lone pairs and two hydrogen atoms, meaning that the total number of bonds of a water molecule is up to four.
Related Question AnswersHow does a hydrogen bond form?
A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. Hydrogen has one proton and one electron. This makes hydrogen an electrically positive atom because it has a deficiency of electrons.Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen.Why are hydrogen bonds important?
Hydrogen bonding is important in many chemical processes. Hydrogen bonding is responsible for water's unique solvent capabilities. Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.What type of bond is a hydrogen bond?
A hydrogen bond is the electromagnetic attraction between polar molecules in which hydrogen is bound to a larger atom, such as oxygen or nitrogen. This is not a sharing of electrons, as in a covalent bond. Instead, this is an attraction between the positive and negative poles of charged atoms.Is ch4 a hydrogen bond?
Answer and Explanation: CH4 cannot form hydrogen bonds. This is because hydrogen bonds are a type of electrostatic interaction, which is only possible in molecules in whichIs HF a hydrogen bond?
The fairly positive hydrogen on one HF molecule will be attracted to one of these lone pairs on a nearby HF molecule. This is a hydrogen bond. Hydrogen bonds are attractions between a δ+ hydrogen on one molecule and a lone pair on a very electronegative atom (N, O or F) on another molecule.Where are hydrogen bonds found?
A ubiquitous example of a hydrogen bond is found between water molecules. In a discrete water molecule, there are two hydrogen atoms and one oxygen atom.Which could accept a hydrogen bond?
The elements that usually participate in hydrogen bonds are nitrogen, oxygen, and fluorine. A hydrogen bond is much weaker than a covalent bond (10-40 kJ/mol vs. The nitrogen atom is called the hydrogen bond acceptor, because it is "accepting" the hydrogen from the oxygen.How do you identify hydrogen?
Burning splint test A splint is lit and held near the opening of the tube, then the stopper is removed to expose the splint to the gas. If the gas is flammable, the mixture ignites. This test is most commonly used to identify hydrogen, which extinguishes with a distinctive 'squeaky pop' sound.What is a hydrogen bond easy definition?
A hydrogen bond is the electromagnetic attraction between polar molecules in which hydrogen is bound to a larger atom, such as oxygen or nitrogen. This is not a sharing of electrons, as in a covalent bond. Instead, this is an attraction between the positive and negative poles of charged atoms.Which hydrogen bond is strongest?
Thus, on a per bond basis, HF H bonding is strongest. However, as water has two H atoms, each molecule can form two H bonds so on a per molecule basis, water H bonding is strongest (this is evidenced by the boiling points of the three substances; NH3 < HF < H2O.)Can hydrogen bonds be easily broken?
Individual hydrogen bonds are weak and easily broken; however, they occur in very large numbers in water and in organic polymers, creating a major force in combination. Hydrogen bonds are also responsible for zipping together the DNA double helix.Do amines form hydrogen bonds?
All of the amines can form hydrogen bonds with water - even the tertiary ones. Although the tertiary amines don't have a hydrogen atom attached to the nitrogen and so can't form hydrogen bonds with themselves, they can form hydrogen bonds with water molecules just using the lone pair on the nitrogen.How do you know which hydrogen bond is stronger?
The strength of an individual H bond depends on the polarity of the H-X bond and therefore on the electronegativity of X ( the more delta positive the H atom, the stronger the electrostatic force of attraction between it and a lone pair of electrons). Thus, on a per bond basis, HF H bonding is strongest.Why are hydrogen bonds so strong?
Hydrogen bonding is a very special intermolecular force that occurs in polar molecules when a hydrogen atom is bonded to a highly electronegative atom. Thus the effect of hydrogen bonds can be very large. They are so strong that they seem like weak covalent bonds. Thus, the name hydrogen bond.What functional groups can hydrogen bond with water?
The Hydroxyl and Carbonyl Groups Oxygen occurs in these two common functional groups: Hydroxyl groups have one hydrogen paired with one oxygen atom (symbolized as -OH). Hydroxyl groups are not highly reactive, but they readily form hydrogen bonds and contribute to making molecules soluble in water.Will ethanol form a hydrogen bond with water?
Yes it can. one ethanol can make hydrogen bond with 3 water molecule theoritically. But practically one ethanol can make hydrogen bond with just 1 water molecule. You now have the complete power and control over the hydrogen bonds in water molecules.What are the 4 types of bonds?
4 Types of Chemical Bonds- 1Ionic bond. Ionic bonding involves a transfer of an electron, so one atom gains an electron while one atom loses an electron.
- 2Covalent bond. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms.
- 3Polar bond.



