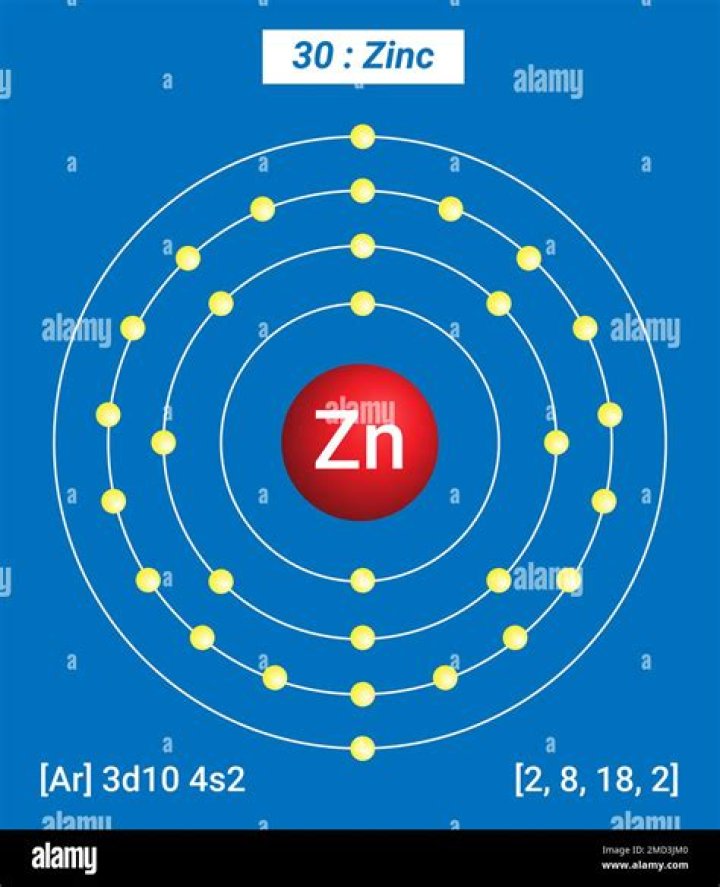
Zn always, to my knowledge, forms a 2+ ion. In fact, all of the elements in that group (Group 12) forms 2+ ions. This is because after losing the 4s electrons Zn still has a a closed n=3 shell, which is a very stable electron configuration..
Beside this, what charge does a zinc ion have?
+2
Also, is Zinc an ionic compound? Chemical properties: Zinc chloride is mostly thought as a ionic compound, however it has been determinate it has a behavior more covalent. The difference of electro-negativity between Zinc and Chloride is 1.3, which is not enough to form a strong ionic bond.
Keeping this in view, why does Zn have a 2+ charge?
Zinc has an electron configuration of [Ar] 3d10 4s2. This small amount of electrons means it is more likely to lose the 2 electrons rather than taking on any in a reaction. Losing all the electrons on the fourth shell would mean Zn lost two negative charges, making it the ion Zn+2.
How many electrons does a zinc ion have?
| Name | Zinc |
| Atomic Mass | 65.39 atomic mass units |
| Number of Protons | 30 |
| Number of Neutrons | 35 |
| Number of Electrons | 30 |
Related Question Answers
Does zinc have a positive or negative charge?
The atomic number of zinc is 30 meaning that its nucleus contains 30 protons. Zinc most commonly forms positively charged cations with a charge of +2. Zinc will rarely form ions with a +1 charge but it will never form ions with a negative charge.What is the difference between Cu and cu2+?
---> Cu is a neutral atom where number of protons and electrons are equal. ----> Cu2+ is a cation formed by lossing 2 electrons. As proton number is more, nuclear Charge for Cu²? will be more and atomic size of Cu²? decreases, Ionization potential is more. So atomic radius of Cu atom is more than Cu2+ ion.Is PB an ion?
So, Na+ is “sodium ion”. For transition metal cations from groups III B – II B (3 -- 12), the cation name is the name of the element, plus the size of the ionic charge. For example, Fe+3 is “iron three ion”. Pb+2 is “lead two ion”.How many bonds can Zinc have?
Chemical reactivity Zinc exhibits only the +2 oxidation state. It can give up two electrons to form an electrovalent compound; e.g., zinc carbonate ZnCO3. It may also share those electrons, as in zinc chloride, ZnCl2, a compound in which the bonds are partly ionic and partly covalent.Is Zinc polar or nonpolar?
ZnZn Bond Polarity
| Electronegativity (Zn) | 1.7 |
| Electronegativity (Zn) | 1.7 |
| Electronegativity Difference | 0 Non-Polar Covalent = 0 0 < Polar Covalent < 2 Ionic (Non-Covalent) ≥ 2 |
| Bond Type | Non-Polar Covalent |
Is Zinc covalent or ionic?
Zinc exhibits only the +2 oxidation state. It can give up two electrons to form an electrovalent compound; e.g., zinc carbonate ZnCO3. It may also share those electrons, as in zinc chloride, ZnCl2, a compound in which the bonds are partly ionic and partly covalent.Is oxygen a cation or anion?
By itself, oxygen is neutral (does not have a charge). In order to be considered a cation or anion, there must be charge involved. Although it actually depends on whether the anode/cathode is considered positive or negative (both can be either, although the other must be the opposite).Is Zn 2+ paramagnetic?
Zn2+ has 0 unpaired d electrons, Co2+ has 3 unpaired d electrons. Co2+ is therefore paramagnetic and will be attracted by a magnetic field.Is Zinc an atom or molecule?
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a blue-silvery appearance when oxidation is removed. It is the first element in group 12 of the periodic table.How many electrons are in the ion Zn 2 +?
The Zn2+ ion has lost two electrons, which leaves it with 30 protons and 28 electrons.Is MgO ionic or covalent?
So 'O'-atom has a tendency to gain two electron ,so that it attain the octet rule . Hence by transfer of two electron from Mg to 'O'-atom , MgO is made of from Mg2+ and O-2 ion . So MgO is ionic compound . It is not a covalent compound.Is zinc soluble in water?
Zinc is insoluble in water but reacts readily with non-oxidising acids, forming zinc (II) and releasing hydrogen. It also dissolves in strong bases. Water solubility of zinc compounds varies, with zinc acetate, zinc nitrate, zinc sulfate, zinc chloride, zinc chlorate and zinc perchlorate all readily soluble in water.Is hydrogen a positive or negative ion?
One Hydrogen (H) has a positive charge +1. This has nothing to do with it having more protons than electrons, as it has one of each. Think of it as 'willing' to donate one electron, or it has +1 electrons see, it is on the plus side. One Oxygen (O) has a negative charge of -2.How are Monatomic cations named?
Monatomic ions that are positively charged ions are called cations. When naming cations, simply take the name of the element and add the term ion to the end. If the cation is formed from a transition metal, give the name of the element followed by the magnitude of the charge written in Roman numerals.Are cations always Metals?
Halogens always form anions, alkali metals and alkaline earth metals always form cations. Most other metals form cations (e.g. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. oxygen, carbon, sulfur).What is the charge on a calcium ion?
The calcium ion, which is a calcium element with a positive charge of 2, is vital to all living creatures. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons.Can zinc and chlorine form ionic bonds?
When forming an ion, a zinc atom loses its two valence electrons, becoming a Zn2+ ion. The chlorine atom has seven valence electrons, and will gain one valence electron to form a chloride ion, Cl1- . Therefore, two chloride ions, Cl1- must bond with one zinc ion, Zn2+ .What is zinc made of?
Zinc is a metal and an element, so it's made out of zinc. It's mostly extracted from the ore Sphalerite, which is zinc sulfide.What type of bond is co2?
Note that carbon dioxide has two covalent bonds between each oxygen atom and the carbon atom, which is shown here as two lines and referred to as a double bond. When molecules are symmetrical, however, the atoms pull equally on the electrons and the charge distribution is uniform.