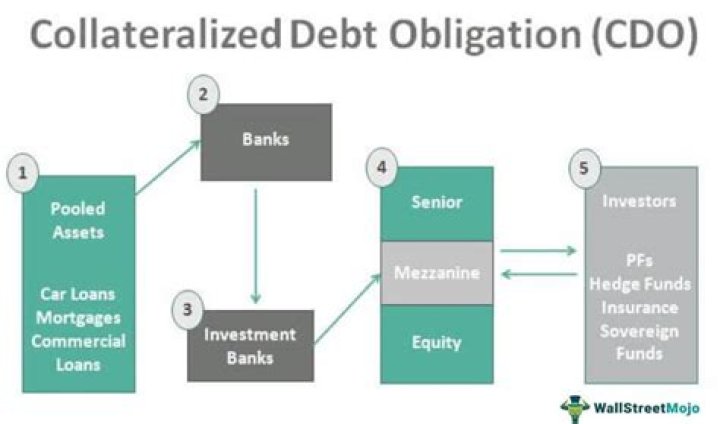
What happens when nitric acid is added to water?
.
Thereof, what happens when hno3 is added to water?
The nitric acid is ionic compound, which is soluble in the water. Since, ionic compound nitric acid is a strong electrolyte. Hence, when nitric acid is dissolved in the water solution then nitric acid dissociates into hydrogen and nitrate ions.
Also, what is the significance of adding nitric acid solution? Nitric acid (HNO3) is added in water samples for heavy metal analysis during the sampling of water. Firstly it reduce the pH of water below 2 and this will reduce the precipitation and adsorption to container wall and it also reduce the bacterial activities. HNO3 is recommended for this because of its oxidizing nature.
People also ask, does red fuming nitric acid react with water?
Soluble in all proportions with water. Dissolution in water produces heat, fumes, and spattering. NITRIC ACID, RED FUMING is a powerful oxidizing agent and nitrating agent. Accelerates the burning of combustible material and may cause charring and then ignition of combustible materials.
Is nitric acid soluble in water?
Nitric acid is a colorless to yellow or red liquid sometimes fuming reddish brown vapors with a suffocating odor. Nitric acid is soluble in water with release of heat.
Related Question AnswersHow do you separate nitric acid from water?
No, we cannot separate water completely from nitric acid. Because water and nitric acid forms azeotropic mixture. Azeotropic mixtures are those mixtures whose boiling point is constant. As a result the components of the mixture boils at the same temperature.What is a common use of nitric acid?
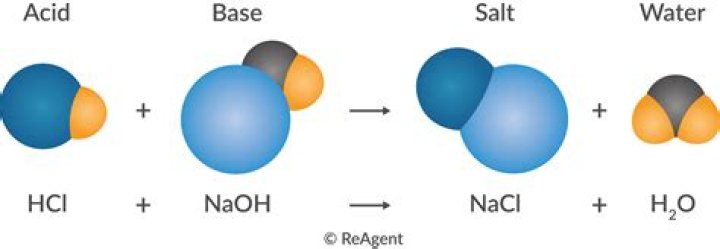
Nitric acid is used in the production of ammonium nitrate for fertilizers, making plastics, and in the manufacture of dyes. It is also used for making explosives such as nitroglycerin and TNT. When it is combined with hydrochloric acid, an element called aqua regia is formed.Is nitric acid strong or weak?
All the other acids are weak. The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid. The only weak acid formed by the reaction between hydrogen and a halogen is hydrofluoric acid (HF).What things contain nitric acid?
Here are the 10 best foods to boost your nitric oxide levels.- Beets. Beets are rich in dietary nitrates, which your body can convert to nitric oxide.
- Garlic.
- Meat.
- Dark Chocolate.
- Leafy Greens.
- Citrus Fruits.
- Pomegranate.
- Nuts and Seeds.
Is nitric acid a compound?
Nitric acid (HNO3), also known as aqua fortis (Latin for "strong water") and spirit of niter, is a highly corrosive mineral acid. The pure compound is colorless, but older samples tend to acquire a yellow cast due to decomposition into oxides of nitrogen and water.What happens when hno2 is dissolved in water?
When HNO2 is dissolved in water it partially dissociates according to the equation HNO2 --> H^+ + NO2^-. A solution is prepared that contains 3.000 g of HNO2 in 1.000 kg of water. Its freezing point is found to be -0.1281 degrees Celsius.What is the equation for nitric acid?
HNO3Why is nitric acid a strong acid?
Nitric acid is a stronger acid than nitrous acid because its conjugate base is more stable. The NO3- ion has excess negative charge spread out over 3 oxygen atoms while the excess negative charge in NO2- is spread out over on 2 oxygen atoms.How do you make fuming nitric acid?
Preparation of nitric acid. Fuming nitric acid is prepared by slowly distilling a mixture of 50 g of sodium nitrate (or equivalent amount of potassium nitrate) and 30 g of concentrated sulfuric acid, in a glass apparatus as shown below. The distillate contains 90-95% of nitric acid.What household products have nitric acid?
Table 7: Alkalis and acids frequently found in household products| Alkalis | Products |
|---|---|
| Acids | Products |
| Sulfuric acid Hydrochloric acid Sodium bisulfate Sodium hypochlorite Acetic Acid Nitric acid | Drain cleaner Toilet bowl cleaner House bleach (low concentration) Descalers |
How do you store fuming nitric acid?
If I were you I would store RFNA and store it in an amber glass bottle with a teflon lined PE lid. Store that bottle in the dark and cool, preferably inside a secondary container. RFNA has a fair amount of dissolved NOx which displaces the equilibrium of nitric acid breakdown to the left.What can nitric acid dissolve?
Nitric acid. Nitric acid is the primary choice here on several grounds. It is an oxidizing acid that can dissolve most metals to form soluble metal nitrates. It has poor oxidizing strength below 2 M but is a powerful oxidizing acid in concentrated form.Why zinc does not react with nitric acid?
Aluminum and zinc do not react with concentrated nitric acid since a thick, hard to dissolve oxidation layer builds (passive oxidation) that protects the metal against further assaults.What is meant by fuming nitric acid?
Definition of fuming nitric acid : concentrated nitric acid containing dissolved nitrogen oxides prepared as either a colorless to pale yellow or a red to brown corrosive poisonous liquid and used especially as a nitrating agent and as a powerful oxidizing agent (as in rocket propellants)How dangerous is nitric acid?
Health Hazards Associated with Nitric Acid Nitric acid is an extremely corrosive acid capable of causing severe chemical burns very rapidly. If nitric acid mists are inhaled, health risks include corrosion of mucous membranes, delayed pulmonary edema, and even death. Contact with eyes can cause permanent cornea damage.What is the PH of nitric acid?
pH of Common Acids and Bases| Acid | Name | 1 mM |
|---|---|---|
| HBr | hydrobromic acid | 3.01 |
| HCl | hydrochloric acid | 3.01 |
| HNO3 | nitric acid | 3.01 |
| H3PO4 | orthophosphoric acid | 3.06 |