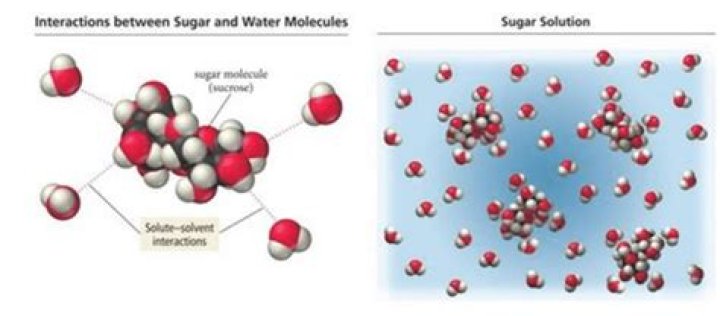
Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. When one of these solids dissolves in water, the ions that form the solid are released into solution, where they become associated with the polar solvent molecules..
In respect to this, how does glucose dissolve in water?
A Water-soluble sugar The reason glucose dissolves readily in water is because it has lots of polar hydroxyl groups which can hydrogen-bond with water molecules. Hydrogen bonds are very important intermolecular forces which determine the shape of molecules like DNA, proteins and cellulose.
Additionally, how much sugar can you dissolve in water? For a given solvent, some solutes have greater solubility than others. For example, sugar is much more soluble in water than is salt. But even sugar has an upper limit on how much can dissolve. In a half liter of 20 °C water, the maximum amount is 1000 grams.
Correspondingly, does glucose dissociate in water?
A common example of a nonelectrolyte is glucose, or C6H12O6. Glucose (sugar) readily dissolves in water, but because it does not dissociate into ions in solution, it is considered a nonelectrolyte; solutions containing glucose do not, therefore, conduct electricity.
Does sugar absorb heat from water?
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water. When water is heated, the molecules gain energy and, thus, move faster. As they move faster, they come into contact with the sugar more often, causing it to dissolve faster.
Related Question Answers
What is chemical formula for glucose?
C6H12O6
What is the chemical formula for sugar dissolved in water?
Because one molecule of water (H2O) is lost in the condensation reaction linking glucose to fructose, sucrose is represented by the formula C12H22O11 (following the general formula Cn[H2O]n - 1).Is starch water soluble?
Starch becomes soluble in water when heated. The granules swell and burst, the semi-crystalline structure is lost and the smaller amylose molecules start leaching out of the granule, forming a network that holds water and increasing the mixture's viscosity. This process is called starch gelatinization.Can sugar form hydrogen bonds with water?
Remembering our definition of hydrogen bond donors and acceptors, you can see that glucose has 5 hydrogen bond donors (H-O) and 6 hydrogen bond acceptors (O). Water molecules can form hydrogen bonds at each of these sites and solvate the glucose molecule. So, it cannot form hydrogen bonds to water.Is sucrose polar or nonpolar?
Sucrose is a polar molecule. The polar water molecules attract the negative and positive areas on the polar sucrose molecules which makes sucrose dissolve in water. A nonpolar substance like mineral oil does not dissolve a polar substance like sucrose.Does water have glucose?
Water will not raise blood glucose levels, which is why it is so beneficial to drink when people with diabetes have high blood sugar, as it enables more glucose to be flushed out of the blood.What is glucose in the body?
Glucose comes from the Greek word for "sweet." It's a type of sugar you get from foods you eat, and your body uses it for energy. As it travels through your bloodstream to your cells, it's called blood glucose or blood sugar. Insulin is a hormone that moves glucose from your blood into the cells for energy and storage.Does glucose ionize in water?
Sugar will readily dissolve in water but doesn't form cations and anions in solution. That is, there are no charge carriers formed. Substances that only partially ionize into ions when dissolved in water are called weak electrolytes. For example, sugar dissolves completely in water but it is a non-electrolyte.How does nacl dissociate in water?
When sodium chloride is dissolved in water, the polar water molecules are able to work their way in between the individual ions in the lattice. The water molecules surround the negative chloride ions and positive sodium ions and pull them away into the solution. This process is called dissociation.Is glucose good for health?
Along with fat, glucose is one of the body's preferred sources of fuel in the form of carbohydrates. People get glucose from bread, fruits, vegetables, and dairy products. You need food to create the energy that helps keep you alive. While glucose is important, like with so many things, it's best in moderation.What is the reaction of glucose?
In respiration, through a series of enzyme-catalysed reactions, glucose is oxidized to eventually to form carbon dioxide and water, yielding energy, mostly in the form of ATP. Chemically joined together, glucose and fructose form sucrose. Starch, cellulose, and glycogen are common glucose polymers (polysaccharides).Why are some ionic compounds insoluble in water?
Generally, ionic compounds whose component ions have larger charge magnitudes are less soluble because the ions are attracted too much to dissociate. A soluble compound will dissociate because of the attractions with the surrounding water molecules are strong enough to separate the ions.What happens when sucrose is added to water?
When granulated sugar is added to water, it breaks apart because the water molecules are attracted to the sucrose molecules through intermolecular forces. As a result, each sucrose molecule is surrounded by water molecules and is carried off into the solution.Does salt ionize in water?
Dissolving a salt molecule in water does not make its atoms ionize. The atoms in solid salts are already ionized long before touching water. Electrons in an atom can only take on specific wave states, and only one electron can occupy one wave state at a time.What happens when salt is dissolved in water?
Water can dissolve salt because the positive part of water molecules attracts the negative chloride ions and the negative part of water molecules attracts the positive sodium ions. The amount of a substance that can dissolve in a liquid (at a particular temperature) is called the solubility of the substance.Why is water called the universal solvent?
Water is capable of dissolving a variety of different substances, which is why it is such a good solvent. And, water is called the "universal solvent" because it dissolves more substances than any other liquid. This allows the water molecule to become attracted to many other different types of molecules.Does salt or sugar dissolve faster in water?
Less sugar is visible in the hot water than in the cold, meaning that more sugar dissolves in the hot water than in the cold water. There is no obvious difference between the amount of salt that dissolves in the hot water compared to the cold water.Which is most soluble in water?
Among given compounds, ethylene glycol ( H O − C H 2 − C H 2 − O H HO-CH_2-CH_2-OH HO−CH2−CH2−OH ) is the most soluble in water. Ethylene glycol has two hydroxy groups both of which form hydrogen bonds with water.Can baking soda be dissolved in water?
Water