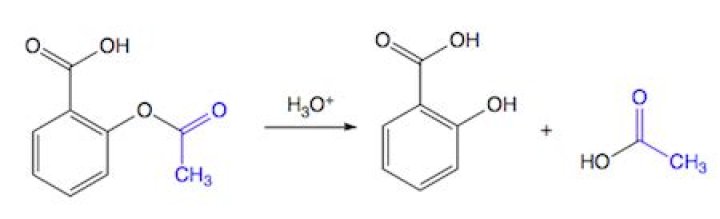
The active ingredient in Aspirin also prevents infections and damage to blood vessels. The efficiency of any drug depends on its chemical stability. Thus, when Aspirin undergoes hydrolysis, the degradation products are salicylic acid and acetic acid. Salicylic acid forms violet-blue complexes with Fe3+ ions..
Also to know is, what is aspirin hydrolysis?
Aspirin can undergo hydrolysis, making it not as effective if it's exposed to water for extended periods of time. The technical name of the active ingredient in aspirin is acetylsalicylic acid. When it reacts with water, we end up with two products, salicylic acid and acetic acid.
Likewise, how does temperature affect the rate of hydrolysis of aspirin? Therefore, temperature does significantly affect the reaction rate of the hydrolysis of Aspirin. Also, as the environment in which the aspirin hydrolysis reaction is occurring becomes more basic, both the rate of reaction of the hydrolysis of aspirin and the formation of salicylic acid increase (Table 1).
In this manner, what happens when aspirin dissolves in water?
Aspirin contains polar functional groups which can form hydrogen bonds with polar water molecules. Ionic salts of aspirin, such as sodium acetylsalicylate, are more soluble in water since they form stronger ion-dipole interactions with water.
How does aspirin break down in the body?
Aspirin is absorbed in the GI tract and then distributed to all tissues of the body. It is broken down into salicylic acid, and the liver changes it into metabolites. Aspirin inhibits the prostaglandin cyclooxygenase, which results in the analgesic, anti-inflammatory, and inhibition of platelet aggregation properties.
Related Question Answers
What does aspirin react with?
The most common drugs that aspirin may interact with are: Anti-inflammatory painkillers, such as diclofenac, ibuprofen, indomethacin, and naproxen. Taken with aspirin, these can increase the risk of bleeding. Warfarin, an anticoagulant drug, or a blood thinner, which stops the blood from clotting.What happens when aspirin is heated?
After the reaction has been heated for some time we add ice to stop it. Aspirin is only slightly soluble in water so if you add it to the reaction mixture the product would rather clump together than be in the water phase. This causes the solid to begin to form or 'precipitate'.Do you dissolve aspirin in water?
? Soluble or dispersible aspirin tablets should be dissolved in a small glass of water before taking. ? Gastro-resistant aspirin tablets have a special coating that helps protect the stomach from the medicine. This type of aspirin tablet can be taken either with or without food.What happens to aspirin in the stomach?
Aspirin is absorbed rapidly from the stomach and intestine by passive diffusion. Aspirin is a prodrug, which is transformed into salicylate in the stomach, in the intestinal mucosa, in the blood and mainly in the liver. Salicylate distributes rapidly into the body fluid compartments. It binds to albumin in the plasma.What happens when you add HCl to aspirin?
Aspirin is a weak acid that also undergoes slow hydrolysis; i.e., each aspirin molecule reacts with two hydroxide ions. To overcome this problem, a known excess amount of base is added to the sample solution and an HCl titration is carried out to determine the amount of unreacted base.How do you measure the rate of hydrolysis of aspirin?
Rate of hydrolysis of aspirin - Measure out 100 cm3 of the pH 7.5 buffer solution into a conical flask.
- Add 0.10 g of powdered aspirin to the buffer solution and stir the mixture gently until the aspirin is completely dissolved.
- Pipette 5 cm3 of iron(III) nitrate reagent directly into a colorimeter tube.
Is aspirin an ester?
The synthesis of aspirin is classified as an esterification reaction. Salicylic acid is treated with acetic anhydride, an acid derivative, causing a chemical reaction that turns salicylic acid's hydroxyl group into an ester group (R-OH → R-OCOCH3).How long does it take for aspirin to dissolve in stomach?
According to the Bayer, a 500 milligram dose of new aspirin starts to work within 16 minutes and brings "meaningful pain relief" within 49 minutes. Regular 500 milligram aspirin takes 100 minutes to do the same.When aspirin is hydrolyzed what are the products?
The efficiency of any drug depends on its chemical stability. Hydrolysis of the drug can be a major reason for the instability of drug solutions. Thus, when Aspirin undergoes hydrolysis, the degradation products are salicylic acid and acetic acid.Which solvent is best for dissolving aspirin?
A stock solution may be made by dissolving the aspirin in the solvent of choice. Aspirin is soluble in organic solvents such as ethanol, DMSO, and dimethyl formamide, which should be purged with an inert gas. The solubility of aspirin in these solvents is approximately 80, 41, and 30 mg/ml, respectively.Does hot water make aspirin tablets dissolve faster?
Hypothesis: The hot water would be the fastest to dissolve the aspirin tablet and cold water would be the slowest. Place 1 aspirin tablet in the cold water and time the aspirin using a stopwatch as soon as the aspirin is in the water and then stop the timer when it completely dissolve.Which brand of aspirin dissolves fastest?
CVS Aspirin dissolved the fastest. The results and data supported the part of my hypothesis that aspirin would dissolve the fastest, but rejected the part of my hypothesis that Bayer would dissolve faster than CVS Aspirin.How do you dissolve Aspirin?
Drop an aspirin tablet into the water (try to avoid splashing – hold the tablet near to the water surface before dropping). Start the stopwatch and immediately withdraw a 1 cm3 sample. Put it into a boiling tube labelled 'zero time'. solution and warm the mixture for 10 minutes in the 70 °C water bath.How long does it take for a tablet to dissolve in water?
After 20-30 minutes, the pills dissolve. Students observe which dissolve the fastest, and discuss the remnants of the various pills.Does the chemical structure of aspirin change when it is dissolved in hot water?
The chemical structure of aspirin did not change when it is dissolved in hot water. The reason adding NaHCO3 to the aspirin crystals is a chemical change is because it forms a new ionic bond with different chemical composition.Why is aspirin not very soluble in water?
Re: solubility of aspirin The reason why it is only partially soluble in water is because of its hydrophobic portion of the molecule. Also, the hydrogen on the carboxylic acid functional group can hydrogen bond with the oxygen atom of the ester carbonyl group making it even further less soluble in water.How do you know if aspirin is decomposing?
It is interesting to note that visually the presence of any whiskers (very thin elongated crystals of salicylic acid) observed on the surface of a solid product con- taining aspirin is definitely an indication that some of the aspirin has decomposed and that the resulting salicylic acid has sublimed through the solidHow does temperature affect aspirin?
As the temperature is increased from −20 °C to 25 °C to 70 °C, the breakage propensity increases and then decreases. The increase in the breakage propensity of Aspirin with increasing temperature is reflected in the values of C1/ρl which increases, corresponding to an increase in H/Kc2.Does temperature affect hydrolysis?
Temperature affects the rates of enzymatic reactions in two ways. Higher temperatures increase the rate of the hydrolysis reaction, but also increase the rate of thermal deactivation of the enzyme.