The reaction is extremely exothermic, producing a bright yellow light and a great deal of heat energy. The hot sodium then reacts with the chlorine, producing a bright yellow light, a great deal of heat energy, and fumes of sodium chloride, which deposits on the walls of the bottle..
In respect to this, does sodium chloride decompose when heated?
Some salts decompose readily upon heating such as bicarbonates salts. While some salts are stable when heated such as sodium chloride. However, if the cation in a salt has a high polarising power towards especially carbonates, hydroxide and nitrates ions, the salt will decompose to form a more stable salt.
Secondly, what happens when you boil sodium chloride? Boiling Point of NaCl When you dissolve salt in water, it breaks into sodium and chloride ions. If you boiled all the water off, the ions would recombine to form solid salt. However, there is no danger of boiling the NaCl: The boiling point of sodium chloride is 2575 F or 1413 C.
In respect to this, what happen when salt is heated?
When common salt is heated the white crystalline solid power remains as it is. Salt powder (solid) does not conduct electricity. But as the temperature is nearly 800 deg. C, the salt reddens and melts into a liquid.
What is the everyday name for sodium chloride?
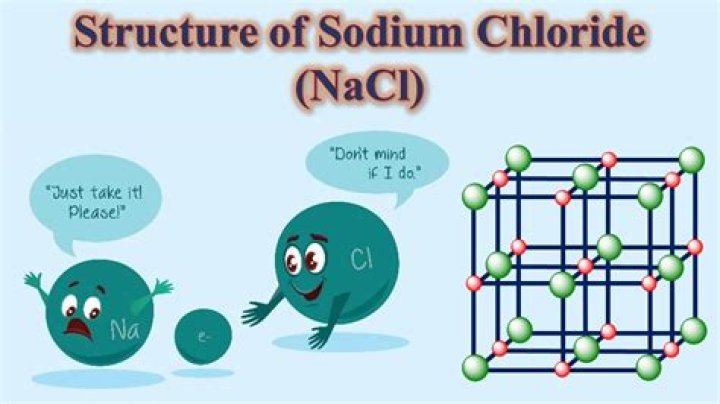
Sodium chloride
| Names |
| IUPAC name Sodium chloride |
| Other names Common salt halite rock salt saline table salt regular salt sea salt |
| Identifiers |
| CAS Number | 7647-14-5 |
Related Question Answers
What are the hazards of sodium chloride?
SYMPTOMS: Ingestion of this compound may cause nausea, vomiting, diarrhea, muscular twitching, inflammation of the gastrointestinal tract, dehydration and congestion (dehydration and congestion can occur in most internal organs, particularly the meninges and brain).How do you get sodium from sodium chloride?
Sodium metal can be obtained from table salt by electrolysis of molten NaCl. In an electrolytic tank,molten NaCl is taken and electrolysed. When electric potential is applied,oppositely charged ions move towards oppositely charged electrodes.What does sodium chloride do?
Sodium chloride is the chemical name for salt. Sodium is an electrolyte that regulates the amount of water in your body. Sodium also plays a part in nerve impulses and muscle contractions. Sodium chloride is used to treat or prevent sodium loss caused by dehydration, excessive sweating, or other causes.Why is the formula for sodium chloride NaCl?
So then, sodium ions will attract chloride ions and form an ionic bond. By the way, chloride is the term used to designate the anion form of chlorine. The chemical formula for sodium chloride is NaCl, which means that for every sodium atom present, there is exactly one chloride atom.What is sodium chloride made of?
Sodium Chloride is a metal halide composed of sodium and chloride with sodium and chloride replacement capabilities.How do you make sodium chloride?
Fill a flask with 0.8 liters of water, weigh out the amount of sodium chloride you need, add it to the water and shake until it dissolves. To make a 1M solution, add 58.44 grams of salt; to make a 0.1M solution, add 5.84 grams; to make a 2M solution, add 116.88 grams and so on.Is sodium chloride toxic to humans?
Human toxicity Consequently, the balance of extracellular and intracellular osmolality is disturbed, which can result in several clinical symptoms, like excessive thirst, weakness, headache, irritability, pulmonary edema, hypertention, tachycardia, and also coma at high NaCl concentrations [1].What happens if you cook salt?
Adding salt at the beginning of cooking gives it time to migrate into the pieces of food, seasoning them throughout. Meanwhile, if you add salt only at the end, it provides a more concentrated, superficial coating that immediately hits your tongue.Can you melt sodium chloride?
Sodium chloride has a high melting and boiling point There are strong electrostatic attractions between the positive and negative ions, and it takes a lot of heat energy to overcome them. Ionic substances all have high melting and boiling points.Can you melt table salt?
Depending on what type of salt (like not table salt), you can melt it more easily than metals. Table salt, however, has a melting temperature of 801 degrees C. Like many other properties, that's less than steel.What does salt do in fire?
Salt is a good means to control/suppress flames and flareups without having a cooling effect on the coals. Possibly they were using it to slow the coals a bit.What happens when you microwave salt?
What happens if you microwave salt? Since the salt cannot absorb much heat, the microwave radiation will damage the microwave, causing it to overheat after a few minutes. Also, it will dry the salt. Since all water will be lost, the salt will collect into solid blocks of salt that are hard to break.What happens when sucrose is heated?
Heating sucrose results in a complex thermal process that involves both melting and decomposition. If there is some water present during the heating process, then a reaction known as sucrose inversion will occur and the sucrose will break down into its components, one molecule of glucose and one molecule of fructose.Why does sodium chloride not melt when heated?
Why doesn't NaCl (sodium chloride) melts when heated with a Bunsen burner flame? The usual explanation is that it has a high melting point due to its ionic bonds. But after some search, i found that the NaCl melting point is around 800º C and the Bunsen burner flame temperature is between 900 and 1,600 °C.Does salt absorb heat?
Salt has an even higher boiling point of 1465 degrees Celsius, or 2669 degrees Fahrenheit. At this temperature, liquid salt turns to vapor. As you can see, salt can absorb tremendous amounts of heat before it undergoes a phase change, turning from solid to liquid and from liquid to vapor.How much heat does sodium chloride absorb?
A large, single crystal of sodium chloride absorbs 98.0 cal of heat. If its temperature rises from 22.0°C to 29.7°C, what is the mass of the NaCl crystal?What happens to the saturated solution of sodium chloride in water upon heating?
Answer and Explanation: When a salt solution is heated, the solvent (assumedly water) will evaporate at a greater rate because the water molecules have greater energy.What happens when salt is added to water?
When salt is mixed with water, the salt dissolves because the covalent bonds of water are stronger than the ionic bonds in the salt molecules. Water molecules pull the sodium and chloride ions apart, breaking the ionic bond that held them together.What does sodium chloride do for the eyes?
Hypertonic sodium chloride eye ointment and eye drops are used to draw water out of a swollen cornea. The extra water in the cornea can cause your vision to be poor. You may also see halos or rings around point light sources. The cornea is the clear cover over the colored part of your eye.