What functional group is present in all phenol compounds?
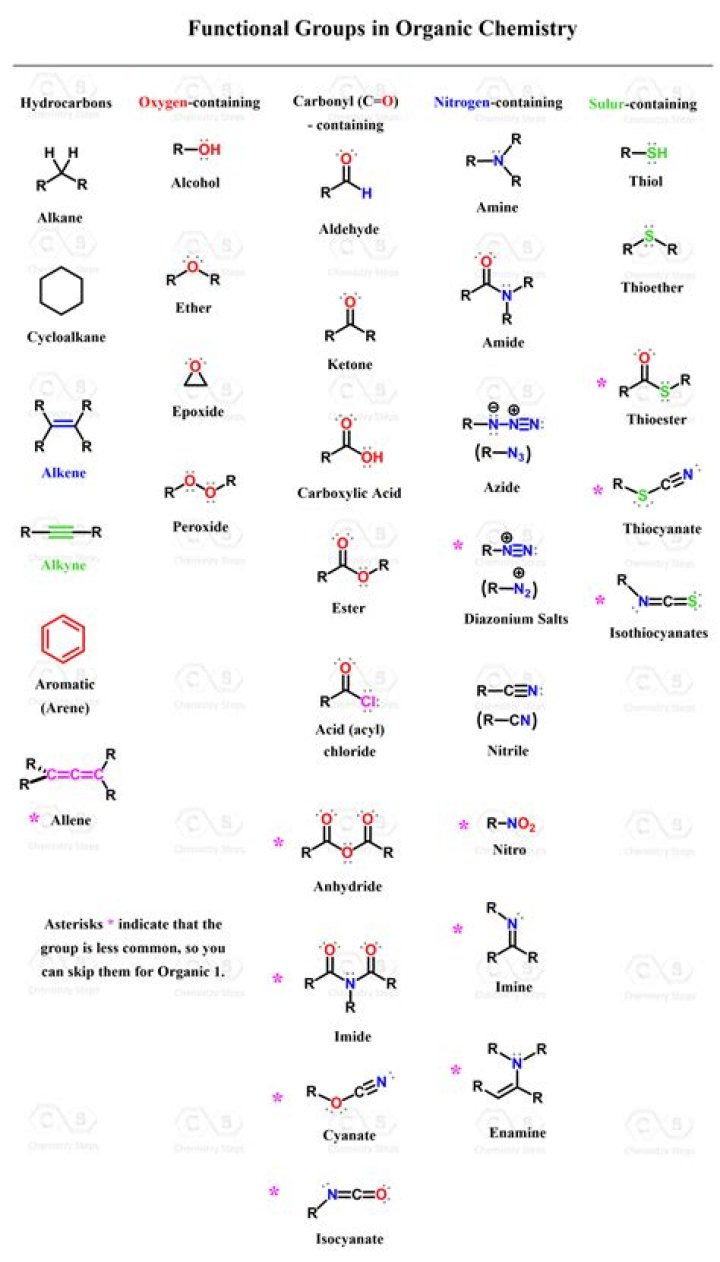
.
Similarly, is phenol a functional group?
Phenyl is a functional group with an aromatic ring bonded to another group. And, phenol is a molecule that is just a phenyl bonded to a hydroxyl group. However, some sources consider phenol itself a functional group.
Also Know, how many different functional groups are present in given compound? A single compound often contains several functional groups. The six-carbon sugar molecules glucose and fructose, for example, contain aldehyde and ketone groups, respectively, and both contain five alcohol groups (a compound with several alcohol groups is often referred to as a 'polyol').
Moreover, what functional group is present in this compound?
carboxylic acid: Any of a class of organic compounds containing a carboxyl functional group—a carbon with one double bond to an oxygen and a single bond to another oxygen, which is in turn bonded to a hydrogen. alcohol: Class of organic compounds containing a hydroxyl functional group.
What is a phenol group?
In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of a hydroxyl group (—OH) bonded directly to an aromatic hydrocarbon group. The simplest of the class is phenol, C. 6H. OH.
Related Question AnswersIs phenol an acid or base?
A hydrogen ion can break away from the -OH group and transfer to a base. For example, in solution in water: Phenol is a very weak acid and the position of equilibrium lies well to the left. Phenol can lose a hydrogen ion because the phenoxide ion formed is stabilised to some extent.How do you identify phenols?
O638: Identification of Phenols – Ferric Chloride Test. Compounds with a phenol group will form a blue, violet, purple, green, or red-brown color upon addition of aqueous ferric chloride. This reaction can be used as a test for phenol groups.Is COOH an acid or base?
Carboxyl groups are weak acids, dissociating partially to release hydrogen ions. The carboxyl group (symbolized as COOH) has both a carbonyl and a hydroxyl group attached to the same carbon atom, resulting in new properties.What is phenol made of?
Phenol is an organic hydroxy compound that consists of benzene bearing a single hydroxy substituent. The parent of the class of phenols. It has a role as a disinfectant, an antiseptic drug, a human xenobiotic metabolite and a mouse metabolite. It is a conjugate acid of a phenolate.Is Arene a functional group?
Ch 11 : Arenes. As a functional group, benzene, substituted benzenes and polybenzenes are called "arenes". Substituted benzenes are usually named as such.What is difference between phenol and phenyl?
Phenol is aromatic hydroxy compound in which hydroxyl group is directly attached to the benzene ring. Whereas phenyl is a monovalent group obtained by replacing one hydrogen atom from benzene.What are simple phenols?
Simple phenols (1) are described as compounds having at least one hydroxyl group attached to an aromatic ring as a basic skeleton. Within the class of simple phenols are phenol (6), catechol (7), resorcinol (8), and phloroglucinol (9). Phenolic acids have a carboxyl group attached to a benzene ring.Is phenol A volatile organic compound?
Phenol is an aromatic organic compound with the molecular formula C6H5OH. It is a white crystalline solid that is volatile. The molecule consists of a phenyl group (−C6H5) bonded to a hydroxy group (−OH). Mildly acidic, it requires careful handling because it can cause chemical burns.What are the 7 functional groups?
There are 7 important functional groups in the chemistry of life: Hydroxyl, Carbonyl, Carboxyl, Amino, Thiol, Phosphate, and aldehyde groups.- Hydroxyl group: consists of a hydrogen atom covalently bonded to an oxygen atom.
- Carbonyl group: is written as a covalent C=O.



