pH, pKa, and Henderson-Hasselbalch Equation The pKa is the pH value at which a chemical species will accept or donate a proton. The lower the pKa, the stronger the acid and the greater the ability to donate a proton in aqueous solution..
Regarding this, when pKa is equal to pH?
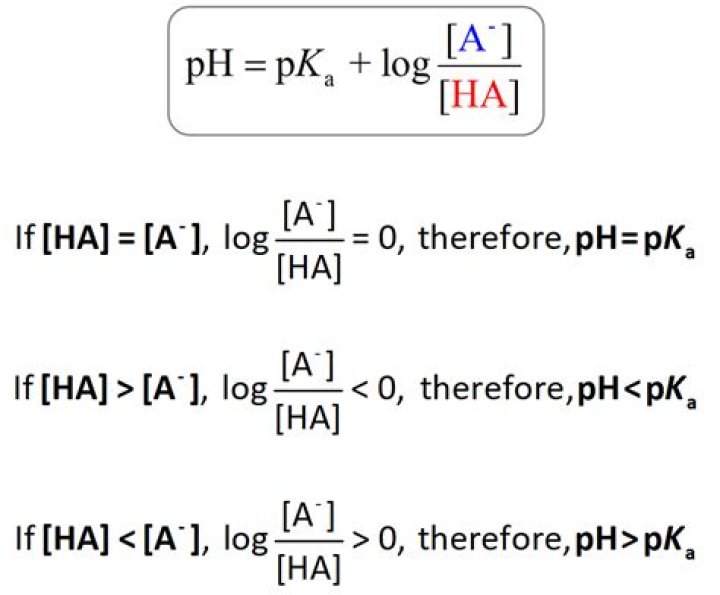
This means that when the pH is equal to the pKa there are equal amounts of protonated and deprotonated forms of the acid. For example, if the pKa of the acid is 4.75, at a pH of 4.75 that acid will exist as 50% protonated and 50% deprotonated.
Subsequently, question is, what effect does increasing the pH have on the value of pKa? Increase in pH Remember that when the pH is equal to the pKa value, the proportion of the conjugate base and conjugate acid are equal to each other. As the pH increases, the proportion of conjugate base increases and predominates.
Hereof, how does KA relate to pH?
Note that x is Related to pH and Calculate Ka Since x = [H3O+] and you know the pH of the solution, you can write x = 10-2.4. It is now possible to find a numerical value for Ka. Ka = (10-2.4)2 /(0.9 - 10-2.4) = 1.8 x 10-5.
What does pKa mean?
Key Takeaways: pKa Definition The pKa value is one method used to indicate the strength of an acid. pKa is the negative log of the acid dissociation constant or Ka value. A lower pKa value indicates a stronger acid. That is, the lower value indicates the acid more fully dissociates in water.
Related Question Answers
What is pKa formula?
pKa is defined as -log10 Ka where Ka = [H+][A-] / [HA]. From these expressions it is possible to derive the Henderson-Hasselbalch equation which is. pKa = pH + log [HA] / [A-] This tells us that when the pH = pKa then log [HA] / [A-] = 0 therefore [HA] = [A-] ie equal amounts of the two forms.What is the pKa of HCl?
Hydrochloric acid (HCl): pKa = -6.3.What happens if pKa is lower than pH?
If the pH is lower than the pKa, then the compound will be protonated. If the pH is higher than the pKa, then the compound will be deprotonated. A further consideration is the charge on the compound. Acids are neutral when protonated and negatively charged (ionized) when deprotonated.How do you convert pKa to pH?
Henderson-Hasselbalch Equation pH = pKa + log [A-]/[HA]. This equation allows you to calculate pH if the dissociation constant, pKa, and the concentrations of the acid and conjugate base are known.Can pKa be negative?
A pKa may be a small, negative number, such as -3 or -5. It may be a larger, positive number, such as 30 or 50. The lower the pKa of a Bronsted acid, the more easily it gives up its proton. The higher the pKa of a Bronsted acid, the more tightly the proton is held, and the less easily the proton is given up.What is the pKa of NaOH?
If NaOH is viewed as a base, its pKa is an ordinary 14 or 15.7.Can pKa change?
The pKa is essentially related to the equilibrium constant. The equilibrium constant is temperature dependent, so changing the temp will change the pKa. The question actually becomes more interesting if you consider moving outside aqueous systems. pKa changes with temperature.How do I calculate pH?
To calculate the pH of an aqueous solution you need to know the concentration of the hydronium ion in moles per liter (molarity). The pH is then calculated using the expression: pH = - log [H3O+].Does higher ka mean stronger acid?
A large Ka value indicates a strong acid because it means the acid is largely dissociated into its ions. A large Ka value also means the formation of products in the reaction is favored. A small Ka value means little of the acid dissociates, so you have a weak acid. The smaller the value of pKa, the stronger the acid.Does higher pKa mean stronger acid?
pKa have an inverse relation with acidic strength. Higher the pKa value weaker the acid. Lower the pKa value stronger the acid is.Does Ka increase with pH?
And if Ka value is decreased, the number of H+ ions in the solution will also be decreased as Ka is directly proportional to [H+] so acidity will decrease. So, if the Pka value is increased, acidic strength decreases and vice versa for basic strength.What is the Ka of a strong acid?
Since the acid dissociates completely, the concentration of products ( [H3O+]and[Cl−] ) is very large and the concentration of the reactant ( [HCl] ) is very small. Therefore, the value of Ka is going to be very large ( ~107 ). The approximate value of pKa is −7 for HCl .What is the difference between pKa and Ka?
Ka is acid dissociation constant and represents the strength of the acid. pKa is the -log of Ka, having a smaller comparable values for analysis. They have an inverse relationship. Larger the Ka, smaller the pKa and stronger the acid.What does Ka tell you about an acid?
The numerical value of Ka is used to predict the extent of acid dissociation. A large Ka value indicates a stronger acid (more of the acid dissociates) and small Ka value indicates a weaker acid (less of the acid dissociates).Is h3po4 an acid or base?
Salts: AgNO3(Salt), NaCl(Salt), NaNO3(salt), KNO3(salt), Acids: H3PO4(acid), HCl(acid), H2SO4(acid), HC2H3O2(acid), HNO3(acid), H2SO3(acid) Bases: Mg(OH)2(base), NaOH(base), KOHbase), NH4OH(base).What is pKa dependent on?
pKa is a measure of acid strength. It depends on the identity and chemical properties of the acid. pH is a measure of [H+] in a solution. For acids, the smaller the pKa, the more acidic the substance is (the more easily a proton is lost, thus the lower the pH).How do you find the pH of a buffer?
To calculate the specific pH of a given buffer, you need to use the Henderson-Hasselbalch equation for acidic buffers: "pH = pKa + log10([A-]/[HA])," where Ka is the "dissociation constant" for the weak acid, [A-] is the concentration of conjugate base and [HA] is the concentration of the weak acid.Does pKa change with concentration?
Let me clarify: pKa does not vary with concentration, as it is related to Ka, an equilibrium constant, which is valid at all concentrations. (Strictly, that should be activities, as activity coefficients vary with concentration.At what volume of added base does pH pKa?
At what volume of added acid does pH=14−pKb? It is at the half-equivalence point when pH=pKa, where pKa=14−pKb. This relationship at the half-equivalence point is described by the Henderson-Hasselbalch equation. At the half-equivalence point, the ratio of weak base over weak acid is equal to 1, and log1=0.