What does e1 stand for in organic chemistry?
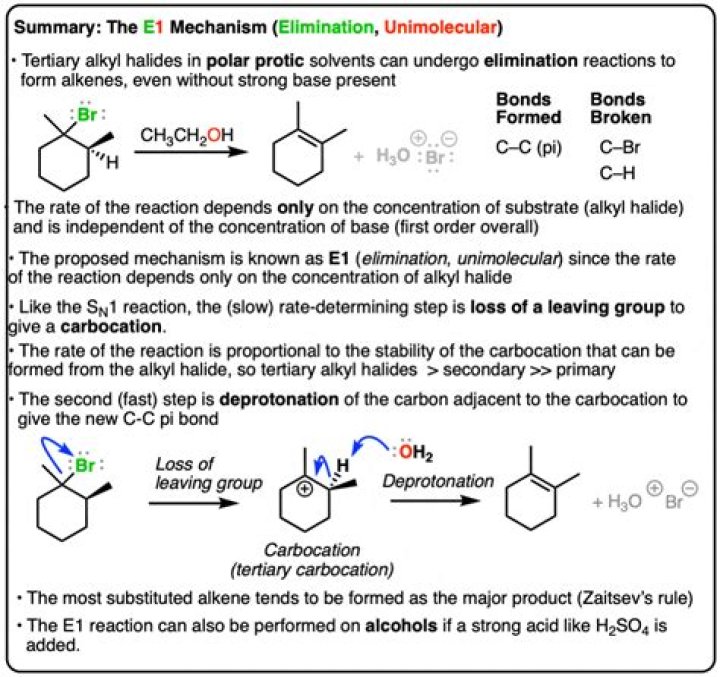
.
Keeping this in consideration, what is e1 in chemistry?
Unimolecular Elimination (E1) is a reaction in which the removal of an HX substituent results in the formation of a double bond. It is similar to a unimolecular nucleophilic substitution reaction (SN1) in various ways. One being the formation of a carbocation intermediate.
Additionally, why are e1 reactions important? Elimination reactions are important as a method for the preparation of alkenes. The term "elimination" describes the fact that a small molecule is lost during the process.
Hereof, what is Solvolysis in organic chemistry?
Solvolysis, a chemical reaction in which the solvent, such as water or alcohol, is one of the reagents and is present in great excess of that required for the reaction. The solvents act as or produce electron-rich atoms or groups of atoms (nucleophiles) that displace an atom or group in the substrate molecule.
Is an e1 reaction concerted?
Hence, the more stable that carbocation is, the faster the reaction will be. The rate of the E2 reaction depends on both substrate and base, since the rate-determining step is bimolecular (concerted). A strong base is generally required, one that will allow for displacement of a polar leaving group.
Related Question AnswersWhat is Saytzeff rule in organic chemistry?
Zaitsev's rule (or Saytzeff's rule, Saytzev's rule) is an empirical rule for predicting the favored alkene product(s) in elimination reactions. More generally, Zaitsev's rule predicts that in an elimination reaction, the most substituted product will be the most stable, and therefore the most favored.Is e1 reversible?
Elimination reactions aren't generally reversible, so products are not directly determined by alkene isomer stabilities. E1 eliminations generally lead to the more stable stereochemistry.What is Alpha elimination?
A 1,1-elimination or α-elimination is an elimination reaction in which an organic compound loses two ligands from the same atom. In this elimination reaction, the two ligands 1 loses, H and Cl, are on the same atom.What is the difference between e1 and e2?
The E1 product is favored by an increase in temperature. In the E2 reaction, we have the same starting compound that is attacked by the base in the first step. An alkene is a major and only product. As we can see, the only difference between these two reactions is a base!What makes a good Nucleophile?
Charge. “The conjugate base is always a better nucleophile”. HO- is a better nucleophile than H2O. The greater the negative charge, the more likely an atom will give up its pair of electrons to form a bond.Why is e1 not stereoselective?
In E1 mechanism, the carbon atom from which the leaving group leaves becomes planer after formation of carbocation (because it is sp2 hybridized). Hence now the only thing that matters for an effective pi-bond formation is- which hydrogen on neighboring c-atom is parallel to the empty orbital on planer carbon atom.Is e1 stereoselective?
E1 reactions are stereoselective – that is when a cis or a trans alkene can be formed, the trans isomer is generally the major product: Remember, trans alkenes are more stable because of the less steric strain.What is alpha and beta elimination?
Elimination reactions are when two groups are removed from a molecule, either simultaneously or sequentially. It is called a “beta elimination” because the carbon the halogen is attached to on old nomenclature was called the alpha (α) carbon and the hydrogen that is eliminated is attached to the neighboring (β) carbon.Is Solvolysis sn1 or sn2?
Solvolysis. Solvolysis is a type of nucleophilic substitution (SN1) /( SN2) or elimination, where the nucleophile is a solvent molecule. Characteristic of SN1 reactions, solvolysis of a chiral reactant affords the racemate.What type of reaction is Solvolysis?
Solvolysis is a type of substitution or elimination reaction in which the solvent acts as a nucleophile. We learned that a nucleophile is anything that can act as an electron pair donor in a chemical reaction and form new bonds with electrophiles, which are electron pair acceptors.What is Hoffman Ammonolysis?
Hoffmann's ammonolysis : In this reaction alkyl halide is treated with alcoholic ammonia solution to give primary amine. If the alkyl halide is in excess, secondary and tertiary amines are formed: Tertiary amine further reacts with alkyl halide to form quaternary ammonium salt.Why is sn1 Unimolecular?
Because the slow step of the reaction involves only the substrate, the reaction is unimolecular. Because only the substrate is present in the transition state, the rate of the reaction depends only on its concentration, and not on the concentration of the nucleophile.What is Nucleophile and Electrophile?
A Nucleophile Is A Reactant That Provides A Pair Of Electrons To Form A New Covalent Bond. An Electrophile Is A Reactant That Accepts A Pair Of Electrons To Form A New Covalent Bond. Nucleophilicity” And “Electrophilicity” Refer To The Extent To Which A Species Can Donate Or Accept A Pair Of Electrons.What is Aminolysis reaction?
Aminolysis (/am·i·nol·y·sis/ amino meaning "contains NH2 group", and lysis meaning "to unbind") is any chemical compound reacts with a molecule of ammonia or an amine and causes a molecule to split into two parts, containing the addition of (or substitution by) an amino group —NH—.Why does racemization occur in sn1?
A) Racemisation occurs in SN1 reaction because in case of SN1 a group (base/nucleophile) attack from( in front and back side) both side. b)in case of second one it has two two acidic hydrogen. thus two path way for this one.What determines Solvolysis rate?
Polar protic solvents actually speed up the rate of the unimolecular substitution reaction because the large dipole moment of the solvent helps to stabilize the transition state. Sometimes in an SN1 reaction the solvent acts as the nucleophile. This is called a solvolysis reaction (see example below).How can you tell the difference between sn1 and sn2 reactions?
The SN1 AND SN2 have few differences they are,- SN1 is unimolecular reaction(first order reaction), SN2 is bimolecular reaction(second order reaction).
- SN2 is stereospecific.
- SN2 depends on nuchleophile and substrate, SN1 depends only on substrate.
- SN2 occur in non-polar solvent.
- SN2 have transition state.



