What do the rows on the periodic table represent?
.
Keeping this in consideration, what do rows tell you on a periodic table?
When you look at the periodic table, each row is called a period (Get it? All of the elements in a period have the same number of atomic orbitals. For example, every element in the top row (the first period) has one orbital for its electrons.
One may also ask, what do the rows on the periodic table have in common? In the periodic table, elements have something in common if they are in the same row. Every element in the top row (the first period) has one orbital for its electrons. All of the elements in the second row (the second period) have two orbitals for their electrons.
Subsequently, question is, what do the rows and columns represent on the periodic table?
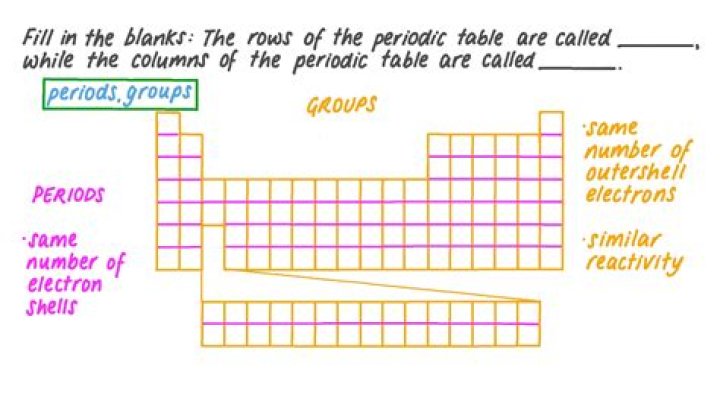
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. All the members of a family of elements have the same number of valence electrons and similar chemical properties. The horizontal rows on the periodic table are called periods.
What does each element in a row represent?
A period in the periodic table is a row of chemical elements. All elements in a row have the same number of electron shells. Each next element in a period has one more proton and is less metallic than its predecessor.
Related Question AnswersWhat are the 7 families of the periodic table?
This list includes alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides, as well as seven elements in groups 3 through 6—aluminum, gallium, indium, thallium, tin, lead, and bismuth.What are the periodic trends in electronegativity?
Electronegativity increases as you move from left to right across a period on the periodic table. This is because, even though there are the same number of energy levels, there are more positive protons in the nucleus, creating a stronger pull on the negative electrons in the outer shell.How many rows are on the periodic table?
sevenWhat is Group 5 called on the periodic table?
Group 5 (by IUPAC style) is a group of elements in the periodic table. Group 5 contains vanadium (V), niobium (Nb), tantalum (Ta) and dubnium (Db). This group lies in the d-block of the periodic table. The group itself has not acquired a trivial name; it belongs to the broader grouping of the transition metals.What can you learn from the periodic table?
The periodic table of elements puts all the known elements into groups with similar properties. This makes it an important tool for chemists, nanotechnologists and other scientists. If you get to understand the periodic table, and learn to use it, you'll be able to predict how chemicals will behave.What is rows and column?
The row is an order in which people, objects or figures are placed alongside or in a straight line. A vertical division of facts, figures or any other details based on category, is called column. Rows go across, i.e. from left to right. On the contrary, Columns are arranged from up to down.What is the difference between rows and columns in the periodic table?
Groups and periods are two ways of categorizing elements in the periodic table. Periods are horizontal rows (across) the periodic table, while groups are vertical columns (down) the table. Atomic number increases as you move down a group or across a period.What does a periodic table tell you?
The atomic number tells you how many protons are in the element, which tells you the number of electrons because they have to balance. The numbers above the periodic table are the group numbers. Elements in the same group have the same number of valence electrons, which means the elements have similar properties.Why is it called the periodic table?
The periodic table got its name from the way the elements are arranged in rows which are called periods. The columns of the table are called groups, some of which have specific names, such as the noble gases and the halogens. Going down the periodic table, the number of atomic orbitals increases by one for each row.How do you describe the periodic table?
The periodic table is a tabular array of the chemical elements organized by atomic number, from the element with the lowest atomic number, hydrogen, to the element with the highest atomic number, oganesson. The atomic number of an element is the number of protons in the nucleus of an atom of that element.Who discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.Why are Group 7 elements called halogens?
Group 7 – The Halogens. The elements in Group 7 of the Periodic Table are all non-metals. They have been given the name halogens from the Greek words meaning 'salt makers'. The salt in your kitchen, sodium chloride, is a compound of the halogen chlorine.How do you figure out the periodic table?
Steps- Get a periodic table of elements.
- Find your element on the periodic table.
- Locate the element's atomic number.
- Determine the number of electrons.
- Look for the atomic mass of the element.
- Subtract the atomic number from the atomic mass.
What is a column called on the periodic table?
The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. The horizontal rows on the periodic table are called periods.What is the significance of the rows and columns in the periodic table?
Rows (Periods) When a column goes from top to bottom, it's called a group. The elements in a group have the same number of electrons in their outer orbital. Those outer electrons are also called valence electrons.What does K stand for on the periodic table?
kaliumHow do you find the group number of an element?
- For s-block elements , group number is equal to the number of valence electrons.
- For p-block elements ,group number is equal to 10+number of electrons electrons in the Valence shell .
- For d-block elements group number is equal to the number of electrons in a (n-1) d subshell + the number of electrons in Valence shell .



