What do covalent and metallic bonds have in common?
.
Thereof, what do ionic covalent and metallic bonds have in common?
Ionic and covalent can be similar bonds. Ionic bonding occurs when transfer of electrons takes place. One atom (or molecule) donates one or more electrons to another. A metallic bond is the attraction between ametal cation and the shared electrons that surround it.
Likewise, do metals have covalent bonding? Metal do form covalent bond. It is very common in transition metal like platinum, palladium. However, it is not the way you are talking about. Typically, when pure metal atoms bond together, they prefer metallic bond.
Regarding this, how are metallic bonds similar to and different from both ionic and covalent bonds?
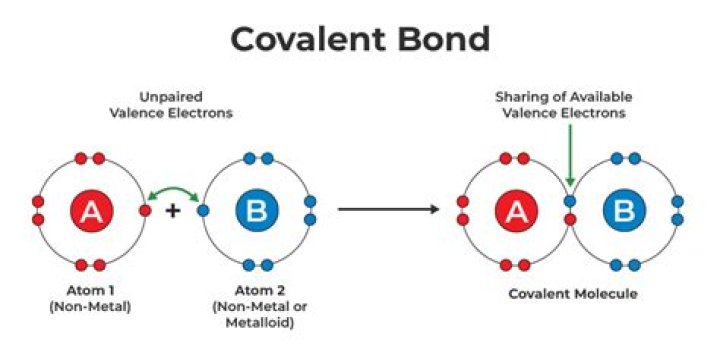
Ionic bonding occurs when transfer of electrons takes place. The ions then attract each other through electrostatic forces of attraction as they are oppositely charged. Covalent bonding occurs when atoms/molecules share pairs of electrons. Metallic bonding is bonding that occurs in metals.
What do metallic and ionic crystals have in common?
While ionic crystals alternate positive charges with negative charges, metallic crystals contain atoms with the same charge surrounded by a sea of electrons. Since these electrons are free to move within the crystal structure, metals are good conductors of electricity and heat.
Related Question AnswersWhat are some examples of ionic bonds?
Ionic bond examples include:- LiF - Lithium Fluoride.
- LiCl - Lithium Chloride.
- LiBr - Lithium Bromide.
- LiI - Lithium Iodide.
- NaF - Sodium Fluoride.
- NaCl - Sodium Chloride.
- NaBr - Sodium Bromide.
- NaI - Sodium Iodide.
Is ionic stronger than metallic?
The metallic bond is somewhat weaker than the ionic and covalent bond. Ionic bonds are strong electrostatic attraction forces formed between positive and negative ions. This bond is non-directional, meaning that the pull of the electrons does not favor one atom over another.Which is the strongest bond ionic covalent or metallic?
The network structure combines to make the substance stronger than normal covalent bonded substances. So to answer your question, substances with standard covalent bonds seem to be weaker than those with ionic bonds because the ionic bonds tend to form a lattice structure, that makes them much stronger.Why are covalent bonds the strongest?
Bond Strength: Covalent Bonds. Stable molecules exist because covalent bonds hold the atoms together. We measure the strength of a covalent bond by the energy required to break it, that is, the energy necessary to separate the bonded atoms. The stronger a bond, the greater the energy required to break it.What is the strongest bond in chemistry?
ionic bondWhich is an example of a metallic bond?
The examples of metallic bond are iron, cobalt, calcium and magnesium, silver, gold, barium, platinum, chromium, copper, zinc, sodium, lithium and francium are some of the examples of metallic bonds.Is NaCl metallic ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.What are the 4 types of bonds?
4 Types of Chemical Bonds- 1Ionic bond. Ionic bonding involves a transfer of an electron, so one atom gains an electron while one atom loses an electron.
- 2Covalent bond. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms.
- 3Polar bond.
What are examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.