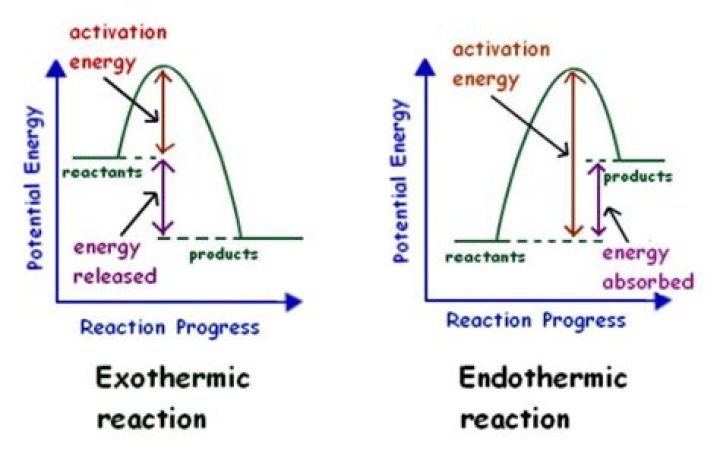
Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and deposition are exothermic processes. Changes of state are examples of phase changes, or phase transitions. All phase changes are accompanied by changes in the energy of a system..
In this regard, what changes are endothermic?
Melting, sublimation and boiling are endothermic reactions -- one that consume energy -- while freezing and condensation are exothermic reactions, which release energy.
Subsequently, question is, is going from a solid to a gas endothermic or exothermic? Phases and Phase Transitions
| Phase Transition | Direction of ΔH |
| Sublimation (solid to gas) | ΔH>0; enthalpy increases (endothermic process) |
| Freezing (liquid to solid) | ΔH<0; enthalpy decreases (exothermic process) |
| Condensation (gas to liquid) | ΔH<0; enthalpy decreases (exothermic process) |
Herein, what are the 4 changes of state?
Substances on Earth can exist in one of four phases, but mostly, they exist in one of three: solid, liquid or gas. Learn the six changes of phase: freezing, melting, condensation, vaporization, sublimation and deposition.
Is boiling exothermic?
Because we must add heat, boiling water is a process that chemists call endothermic. Clearly, if some processes require heat, others must give off heat when they take place. These are known as exothermic. Likewise when liquid water freezes, heat is given off.
Related Question Answers
What phase change is endothermic?
Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and deposition are exothermic processes. Changes of state are examples of phase changes, or phase transitions. All phase changes are accompanied by changes in the energy of a system.Is ice melting endothermic?
Well, it's a little easier going the other way. Melting ice is endothermic -- you can see this by putting a thermometer in a glass of warm water, adding an ice cube, and watching the temperature go down as the ice melts. The melting process needs heat to proceed and takes it from the warm water.Is exothermic hot or cold?
In an exothermic reaction, the system loses heat as the surroundings heat up. That ice pack the coach is putting on the batter? It's an example of an endothermic reaction. An endothermic reaction is when heat is needed by the reaction, so it draws heat from its surroundings, making them feel cold.What is an example of endothermic change?
These examples could be written as chemical reactions, but are more generally considered to be endothermic or heat-absorbing processes: Melting ice cubes. Converting frost to water vapor (melting, boiling, and evaporation, in general, are endothermic processes. Making an anhydrous salt from a hydrate.Are humans endothermic?
You probably know that humans are warm-blooded, while creatures like snakes are cold-blooded. Humans, on the other hand, are endothermic which means our body chemistry regulates our temperature and keeps it constant.Why is freezing exothermic?
Freezing, the phase transition from liquid to solid form, is an exothermic process because energy, in the form of heat, is emitted in the process. Because freezing/melting is a first-order phase transition, there is a latent heat involved in the transition.What is the example of exothermic reaction?
Another simple example of an exothermic reaction is combustion, such as lighting a candle. An initial input of energy causes oxygen and wax react to produce carbon dioxide, water, and heat.What causes change of state?
Changes of state are physical changes in matter. They are reversible changes that do not change matter's chemical makeup or chemical properties. Processes involved in changes of state include melting, freezing, sublimation, deposition, condensation, and evaporation. Energy is always involved in changes of state.What happens during change of state?
During a change in state the heat energy is used to change the bonding between the molecules. In the case of melting, added energy is used to break the bonds between the molecules. Below is a picture of solid ice melting into liquid water.Is boiling a change of state?
The boiling point of a substance is the temperature at which it can change its state from a liquid to a gas throughout the bulk of the liquid. Any change of state from a liquid to a gas at boiling point is considered vaporization.What is the definition of change of state?
1. state change - a change from one state (solid or liquid or gas) to another without a change in chemical composition. phase change, phase transition, physical change. freeze, freezing - the withdrawal of heat to change something from a liquid to a solid. liquefaction - the conversion of a solid or a gas into a liquid.What do you mean by change of state?
change of state. The physical process where matter moves from one state to another. Examples of such changes are melting, evaporation, boiling, condensation, freezing, crystallization, and sublimation.How many states of matter are there?
The five phases of matter. There are four natural states of matter: Solids, liquids, gases and plasma. The fifth state is the man-made Bose-Einstein condensates. In a solid, particles are packed tightly together so they don't move much.What is an example of a change of state?
Phase changes include vaporization, condensation, melting, freezing, sublimation, and deposition. Evaporation, a type of vaporization, occurs when particles of a liquid reach a high enough energy to leave the surface of the liquid and change into the gas state. An example of evaporation is a puddle of water drying out.What is the 7 states of matter?
Erevything around us is made of matter. Matter is any thing that is made from atoms and molecules.( Studios, 1995) .The seven states of matter that I am investigating are Solids, Liquids, Gases, Ionized Plasma, Quark-Gluon Plasma, Bose-Einstein Condensate and Fermionic Condensate.Why changes of state are physical changes?
When matter changes from one state to another, the substance itself does not change. Water, ice, and water vapor are all the same basic substance. Changes in state are physical changes because changes in state do not change the basic substance.Is a fridge endothermic or exothermic?
Nope it is a machine that uses the principles of expansion of a liquid to a gas. This makes the refrigerator cold in side. An endothermic reaction is one than causes a temperature drop as the reaction absorbs energy from its surroundings.How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.Which process is exothermic?
In thermodynamics, the term exothermic process (exo- : "outside") describes a process or reaction that releases energy from the system to its surroundings, usually in the form of heat, but also in a form of light (e.g. a spark, flame, or flash), electricity (e.g. a battery), or sound (e.g. explosion heard when burning