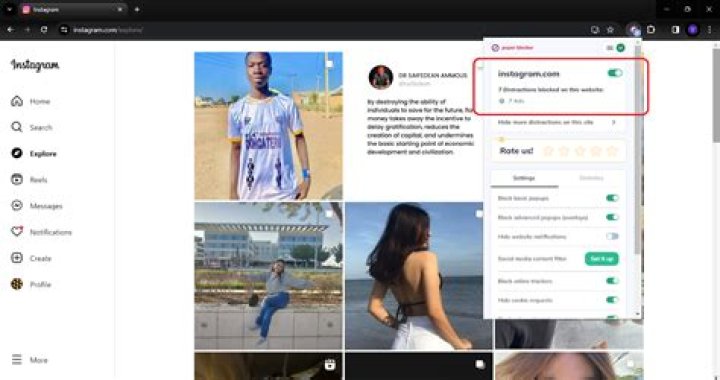
the SI unit of kilogram per cubic metre (kg/m3) and the cgs unit of gram per cubic centimetre (g/cm3) are probably the most commonly used units for density..
Hereof, what is the basic SI unit for density?
kilogram
Also Know, what is the SI unit of relative density? The SI unit of relative density is kg/m 3.
Accordingly, what are the correct units for density?
An introduction to density Density is the mass of an object divided by its volume. Density often has units of grams per cubic centimeter (g/cm3). Remember, grams is a mass and cubic centimeters is a volume (the same volume as 1 milliliter).
What are the basic SI units?
There are seven basic units in the SI system: the meter (m), the kilogram (kg), the second (s), the kelvin (K), the ampere (A), the mole (mol), and the candela (cd).
Related Question Answers
What is the SI unit for energy?
joules
How do you convert SI units?
1 Answer. Multiply or divide by powers of 10. SI units are easy to convert because you multiply or divide by 10 - sometimes more than once. Think of a staircase - every time you step up a stair, you divide by 10; every time you step down a stair, you multiply by 10.Why do we need SI units?
The SI system is important in science and technology because it provides a consistent and coherent system of units for all physical quantities, derived from a few base units. This means that you do not need to use any sort of conversion factor between different units;.What is the SI unit for area?
Another example of a derived SI unit for measuring area is meter square (m2). To find the area, we multiply measurement of length and the measurement of width. In this example, the same SI unit is multiplying to each other, giving us meter square (m2).Is liter an SI unit?
The litre (British, Commonwealth & French spelling) or liter (American, Scandinavian, German & Filipino spelling) (SI symbols L and l, other symbol used: ℓ) is a non-SI unit of volume. It is equal to 1 cubic decimetre (dm3), 1,000 cubic centimetres (cm3) or 0.001 cubic metre.Is density measured in kilograms?
Density is the mass per unit volume of a substance. density = mass/volume. The unit for mass is kilograms (kg)/grams (g) and volume in centimetres cubed (cm3) or metres cubed (m3).What is another name for density?
density, denseness(noun) the amount per unit size. Synonyms: tightness, compactness, slow-wittedness, dumbness, denseness, concentration. concentration, density, denseness, tightness, compactness(noun)How can I calculate density?
To find the density of any object, you need to know the Mass (grams) of the object, and its Volume (measured in mL or cm³). Divide the mass by the volume in order to get an object's Density.Does density affect weight?
Density is merely the ratio of the mass of an object to its volume. Density affects “weight” (or gravitational forces felt by the object itself / other objects), but only in the manner described above (i.e. it all has to do with the mass spatial distribution, which is exactly what density “quantifies”, in a way).Does density vary with temperature?
Density changes with temperature because volume changes with temperature. Density is mass divided by volume. As you heat something up, the volume usually increases because the faster moving molecules are further apart. Since volume is in the denominator, increasing the volume decreases the density.How does density work?
Density is a measure of how compact the mass in a substance or object is. The density of an object or substance can be calculated from this equation: density in kilograms per meter cubed is equal to mass in kilograms, divided by volume in meters cubed. Or in other words, density is mass spread out over a volume.What is the density of solid?
The ratio of the mass of a substance to its volume is known as the mass density or, simply, the density of the substance. The volume of an irregularly-shaped solid can be measured by submersion in a liquid — the difference in volume caused by addition of the solid is equal to the volume of the solid.Why do we measure density?
Density is important because it affects whether objects will float or sink. It is an important property to consider when building things like ships and hot air balloons. He realized that the amount of water that spilled was equal in volume to the space that his body occupied.Which material has highest density?
osmium
What is specific gravity units?
Specific gravity is a dimensionless quantity; that is, it is not expressed in units. To find the sp gr of a solid or liquid, you must know its density in kilograms per meter cubed (kg/m3) or in grams per centimeter cubed (g/cm3). Then, divide this density by the density of pure water in the same units.What is specific gravity of water?
The specific gravity of an object is the ratio between the density of an object to a reference liquid. Usually, our reference liquid is water, which has a density of 1 g/mL or 1 g/cm^3.What is specific gravity of blood?
Blood normally has a specific gravity of approximately 1.060.Why is specific gravity important?
4.1 Specific gravity is an important property of fluids being related to density and viscosity. Knowing the specific gravity will allow determination of a fluid's characteristics compared to a standard, usually water, at a specified temperature.What is specific gravity of soil?
The "specific gravity" of soils and soil solids refers to the mass of solids in the soil compared to the mass of water at the same volume. In general, the specific gravity tends to fall between 2.65 and 2.80, with coarser soils generally having lower specific gravities than finer soils.