What are the FDA regulations for dietary supplements?
.
Keeping this in consideration, are dietary supplements regulated by the FDA?
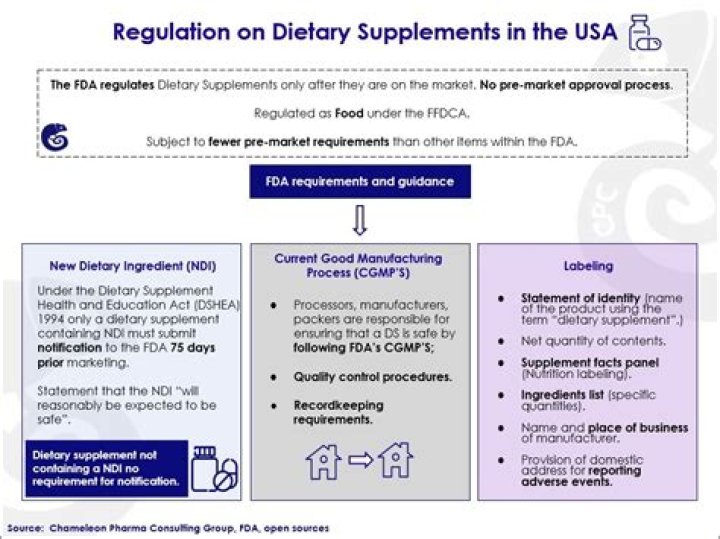
Dietary Supplements. FDA regulates both finished dietary supplement products and dietary ingredients. FDA regulates dietary supplements under a different set of regulations than those covering "conventional" foods and drug products. Under the Dietary Supplement Health and Education Act of 1994 (DSHEA):
Additionally, what are the regulations for supplements? Herbal supplements are regulated by the FDA, but not as drugs or as foods. They fall under a category called dietary supplements. The rules for dietary supplements are as follows: Manufacturers don't have to seek FDA approval before selling dietary supplements.
Keeping this in view, what are the current regulations in the United States regarding dietary supplements?
Federal Regulation of Dietary Supplements The U.S. Food and Drug Administration (FDA), which regulates dietary supplements, requires that companies submit safety data about any new ingredient not sold in the United States in a dietary supplement before 1994.
Do you need FDA approval to sell supplements?
Federal law does not require dietary supplements to be proven safe to FDA's satisfaction before they are marketed. Dietary supplement manufacturers do not have to get the agency's approval before producing or selling these products.
Related Question AnswersWhat are some examples of dietary supplements?
Dietary supplements come in a variety of forms, including tablets, capsules, gummies, and powders, as well as drinks and energy bars. Popular supplements include vitamins D and B12; minerals like calcium and iron; herbs such as echinacea and garlic; and products like glucosamine, probiotics, and fish oils.Are GNC products safe?
First, GNC has agreed that, upon learning that the FDA has issued a public written notice indicating that a purported dietary supplement or an ingredient contained in a purported dietary supplement is not legal and/or not safe, GNC will take immediate action to suspend the sale of such a product or products.Do we really need supplements?
Most people do not need to take vitamin supplements and can get all the vitamins and minerals they need by eating a healthy, balanced diet. Vitamins and minerals, such as iron, calcium and vitamin C, are essential nutrients that your body needs in small amounts to work properly.What supplements should you not take together?
5 Nutrients to Take – or Avoid Taking – Together for Best Absorption- Nutrient Absorption Guidelines.
- Pair: Iron + Vitamin C.
- Pair: Vitamin D + Calcium.
- Pair: Vitamin B12 + Folate.
- Pair: Vitamin D + Omega-3s.
- Avoid: Iron + Calcium.
How do I know if a supplement is FDA approved?
To find out if a drug is approved by FDA, consumers can use two different Internet sites:- [email protected] lists most prescription and over-the-counter (OTC) drug products approved since 1939.
- The FDA "Orange Book" contains drugs approved by FDA under New Drug Applications (NDAs) and Abbreviated New Drug Applications (ANDAs).
Is advocare FDA approved?
* It carries no warning from the Food and Drug Administration (FDA) and is approved by the FDA as well as the World Health Organization Expert Committee on Food Additives. Additionally, it is approved by food and health agencies of over 40 countries.Who needs dietary supplements?
Examples of population groups requiring specific advice about supplements| Population group | Nutrients |
|---|---|
| Women of childbearing age | Folic acid and vitamin D, possibly iron |
| Children under age 5 | Vitamin A, vitamin C, vitamin D, although children with a good appetite who eat a wide variety of food may not need them. |
Is c4 FDA approved?
Popular pre-workout supplements on the market today include products such as Jack3d, C4 Extreme and N.O-Xplode. However, all contain the non-FDA approved dietary supplement methylhexanamine (listed as 1,3-dimethylamylamine on product labels).What are the side effects of taking supplements?
Common–But Surprising–Side Effects of 6 Popular Supplements- Calcium carbonate and constipation.
- Magnesium citrate and diarrhea.
- Vitamin D and kidney stones.
- Vitamin E—and omega-3s—and blood thinning.
- Vitamin B complex and anxiety.
- Zinc and nausea.
How do I know if my supplements are good quality?
Are Your Supplements Safe? Here's How to Tell- Make sure you need the supplement.
- Check for supporting science.
- Speak with your doctor if you are taking medication, pregnant, or breastfeeding.
- Buy supplements from a reputable source.
- Look for a third party verification on the bottle.
- Look for third party verification online.
- Buy organic herbal supplements.
What supplements are proven to work?
Here are five supplements health experts actually use.- Fish oil. "One of the key nutrients many of us don't get enough of is long chain omega 3 fats (which are found naturally in oily fish, for example, salmon)," Debenham told HuffPost Australia.
- Probiotics.
- Vitamin D.
- Magnesium.
- Protein.
What is required to get a supplement approved for sale in the US?
A dietary supplement is considered “new” if it contains an ingredient not recognized as a food substance, unless it was sold as a supplement before October 1994. If it is new, the manufacturer must provide the FDA with reasonable evidence that the new ingredient is safe before the supplement is marketed to the public.What is the difference between vitamins and supplements?
The actual difference between vitamins and supplements: “Supplements are any product that adds nutritional value to your diet or augments health. Vitamins are a type of supplement. Other types of supplements include minerals, herbs, or nutrients like fiber.”How many dietary supplements are on the market today?
More than half of American adults take at least one dietary supplement a day. And the number of these products on the market is growing—from about 4,000 in 1994 to about 80,000 today, for an estimated $40 billion in sales.Are supplements safe?
Are there any risks in taking supplements? Yes. Many supplements contain active ingredients that have strong biological effects in the body. This could make them unsafe in some situations and hurt or complicate your health.Is Nature's Way FDA approved?
WASHINGTON--The Food and Drug Administration (FDA) issued a warning letter to Nature's Way in late March. According to AHPA, the FDA states that, as labeled, the product "presents a significant or unreasonable risk of injury or illness." Visit www.fda.gov for more information.What is the best weight loss supplement?
Here are the 12 most popular weight loss pills and supplements, reviewed by science.- Garcinia Cambogia Extract. Share on Pinterest.
- Hydroxycut.
- Caffeine.
- Orlistat (Alli)
- Raspberry Ketones.
- Green Coffee Bean Extract.
- Glucomannan.
- Meratrim.



