carbon - a nonmetallic chemical element found in many inorganic compounds and all organic compounds: diamond and graphite are pure carbon; carbon is the basic element in coal, coke, charcoal, soot, etc.: symbol, C; at.
- a sheet of carbon paper.
- carbon copy.
- a stick of carbon used in an arc lamp.
.
Just so, what are some examples of carbon?
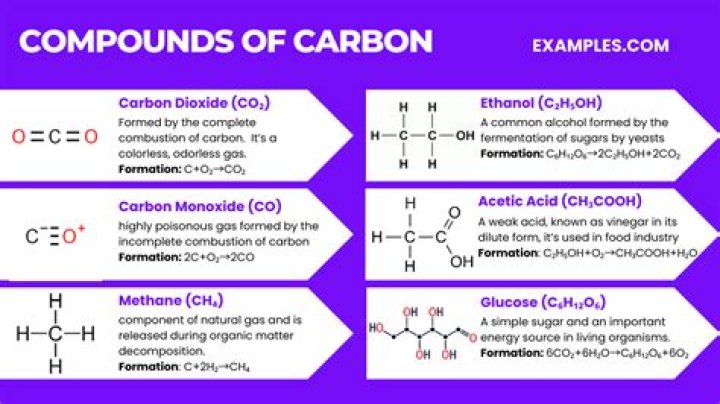
Carbon forms compounds such as methane, propane, butane, benzene and carbon dioxide. Silicon forms compounds such as silane, disilicon hexahydride, silicon tetrafluoride and silicon tetrachloride. Carbon and silicon are often combined, with carbon serving as silicon's backbone.
Subsequently, question is, how is carbon used in everyday life? Uses of Carbon in daily life
- It makes up for 18% of the human body. Sugar, glucose, proteins etc are all made of it.
- Carbon in its diamond form is used in jewellery.
- Amorphous carbon is used to make inks and paints.
- Graphite is used as the lead in your pencils.
- One of the most important uses is carbon dating.
People also ask, is carbon An example of an element?
Carbon is the sixth element in the periodic table. Located between boron (B) and nitrogen (N), it is a very stable element. Because it is stable, it can be found both by itself and in many naturally occurring compounds. Scientists describe the three states of carbon as diamond, amorphous, and graphite.
What everyday items contain carbon?
There are three common naturally occurring forms of carbon: graphite, amorphous carbon, and diamond. These are used in many modern products including inks, rubber, steel, pencils, and more! Tens of millions of artificial carbon compounds are useful for petroleum (gasoline) and plastics.
Related Question Answers
Are humans made of carbon?
Almost 99% of the mass of the human body is made up of six elements: oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. Only about 0.85% is composed of another five elements: potassium, sulfur, sodium, chlorine, and magnesium. All 11 are necessary for life.Do humans contain carbon?
Carbon, the basic unit for organic molecules, comes in second. 99% of the mass of the human body is made up of just six elements: oxygen, carbon, hydrogen, nitrogen, calcium, and phosphorus. Carbon (C) - 18% - Carbon has four bonding sites for other atoms, which makes it the key atom for organic chemistry.What is the importance of carbon?
Carbon is Important to Life In its metabolism of food and respiration, an animal consumes glucose (C6H12O6), which combines with oxygen (O2) to produce carbon dioxide (CO2), water (H2O), and energy, which is given off as heat. The animal has no need for the carbon dioxide and releases it into the atmosphere.What is oxygen made of?
Under standard conditions oxygen forms a gas that is composed of molecules consisting of two oxygen atoms (O2). This is called a diatomic gas. In this form oxygen is a colorless, odorless, tasteless gas. Oxygen also exists as the allotrope ozone (O3).How do you melt carbon?
Note: At normal atmospheric pressure, carbon does not melt when heated, it sublimes. i.e. it undergoes a phase change directly from solid to gas. If the pressure is increased to 10 atmospheres carbon (graphite) is observed to melt at 3550 °C.What are 4 properties of carbon?
Carbon has several allotropes, or different forms in which it can exist. These allotropes include graphite and diamond, which have very different properties. Despite carbon's ability to make 4 bonds and its presence in many compounds, it is highly unreactive under normal conditions.How are we made of carbon?
As well as being the main element in organic matter, carbon atoms are the only element in both graphite and diamond. It would be impossible for life on earth to exist without carbon. Carbon is the main component of sugars, proteins, fats, DNA, muscle tissue, pretty much everything in your body.What is the color of carbon?
The physical properties of carbon varies depending on its allotropic form; diamond is a colorless, hard crystalline solid, graphite is soft, black and shiny, and graphene, fullerenes and nano-forms look like dark brown or black soot-like powders. All the allotropes of carbon are solids under normal conditions.Is Salt an element?
Chemically, table salt consists of two elements, sodium (Na) and chloride (Cl). Neither element occurs separately and free in nature, but are found bound together as the compound sodium chloride.Why is carbon a non metal?
Carbon doesn't contain the physical properties of a metal. Most metals are usually lustrous, opaque, solid, and some are malleable and ductile. Metals are also very good conductors. In this case, carbon is a non metal.Is oxygen a metal?
Oxygen is not a metal. The periodic table is separated into three parts: metals, semi-metals (which are also known as mateliods) and non-metals. Oxygen classifies as a non metal. It is a gas at room temperature, cannot conduct electricity and has a boiling point of -182.96ºC and a melting point of -218.79ºC.What family is carbon in?
The carbon family consists of the elements carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl). Atoms of elements in this group have four valence electrons. The carbon family is also known as the carbon group, group 14, or the tetrels.Is Sugar an element?
The white stuff we know as sugar is sucrose, a molecule composed of 12 atoms of carbon, 22 atoms of hydrogen, and 11 atoms of oxygen (C12H22O11). Like all compounds made from these three elements, sugar is a carbohydrate. Sucrose is actually two simpler sugars stuck together: fructose and glucose.Is coal a carbon?
Coal is a combustible black or brownish-black sedimentary rock, formed as rock strata called coal seams. Coal is mostly carbon with variable amounts of other elements; chiefly hydrogen, sulfur, oxygen, and nitrogen.When was carbon found?
Discovered: Known since ancient times. It was first recognized as an element in the second half of the 18th century. Name: A.L. Lavoisier proposed carbon in 1789 from the Latin carbo meaning "charcoal." A.G. Werner and D.L.G.What is graphite made of?
Graphite is made of pure carbon. Carbon atoms are capable of forming bonds which create a number of different structures. Diamond and graphite are two of the most well-known forms (allotropes) of carbon.Where is carbon found in the human body?
It is the fourth most abundant element in the universe, the fifteenth most abundant element on Earth, and the second most abundant element in the human body, after oxygen. Carbon is present in all known life forms. It can be found dissolved in all water bodies on the planet. Most of it is stored in rocks.What are the 2 properties of carbon?
The two properties that give rise to carbon to form a large number of compounds are: 1) Catenation: It is the ability to form bonds with other carbon atoms. 2) Tetravalency: Due to 4 valency of carbon, it is capable of bonding with four other atoms.Is Salt a carbon compound?
NO, the salt that we use in daily life is not a carbon compound. The chemical formula of the common salt that we use every day is NaCl. The symbol for the element carbon is C. There is no carbon in the chemical formula for salt.