What are Lewis structures and how are they involved in chemical bonds?
.
Likewise, people ask, what electrons are involved in chemical bonding and are represented by Lewis structures?
When different atoms interact to form a bond, only their outermost regions come in contact; only the valence electrons are involved. Lewis dot symbol: contains the symbol of an element and one "dot" for each valence electron in an atom; used to represent the distribution of valence electrons in a molecule.
what is Lewis rule? Lewis Dot Rules. In 1916, ten years before the Schrodinger wave equation, G. N. Lewis suggested that a chemical bond involved sharing of electrons. He described what he called the cubical atom , because a cube has 8 corners, to represent the outer valence shell electrons which can be shared to create a bond.
Keeping this in consideration, how does the octet rule apply to chemical bonding?
The octet rule can be used to explain the number of covalent bonds an atom forms. This number normally equals the number of electrons that atom needs to have a total of eight electrons (an octet) in its outer shell. For example, the halogens (Group VIIA), all have seven valence electrons.
What are the two main types of chemical bonds?
There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds. Atoms that share electrons in a chemical bond have covalent bonds. An oxygen molecule (O2) is a good example of a molecule with a covalent bond.
Related Question AnswersWhat electrons are involved in the formation of chemical bonds?
Preview Flashcards| Front | Back |
|---|---|
| The electrons involved in the formation of a chemical bond are called | Valence electrons. |
| A chemical bond that results from the electrostatic attraction between positive and negative ions is called | An ionic bond. |
What is meant by chemical bond?
A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds.Why are bonding theories important?
Why are bonding theories important? Provide some examples of what bonding theories can predict. Bonding theories are central to chemistry because they explain how atoms bond together to form molecules. Chemical bonds form because they lower the potential energy between the charged particles that compose the atom.What is the definition of structural formula in chemistry?
The structural formula of a chemical compound is a graphic representation of the molecular structure (determined by structural chemistry methods), showing how the atoms are possibly arranged in the real three-dimensional space. The chemical bonding within the molecule is also shown, either explicitly or implicitly.What property determines what kind of bond is formed between two elements?
electronegativityWhat is the Lewis structure for h2o?
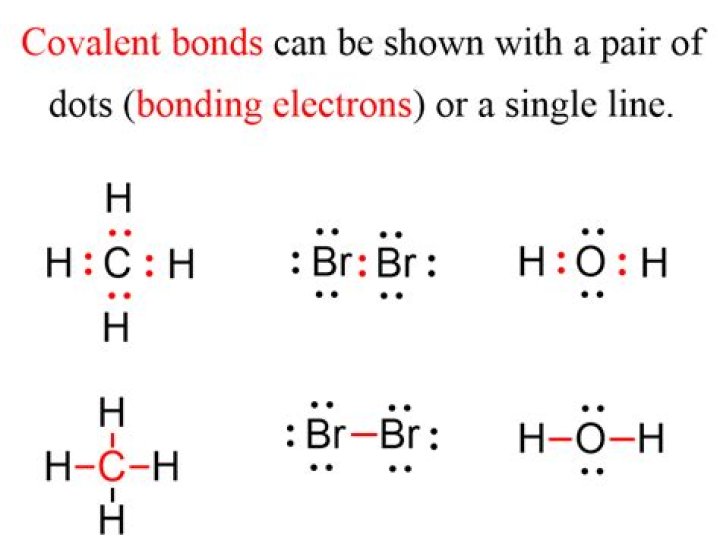
The skeleton structure is H-O-H. O has 6 valence electrons, and each H has one. You must arrange 8 electrons in pairs so that O has 8 and each H has two electrons in its valence shell. You have eight valence electrons in your trial structure, so it has the correct number of electrons.How do you draw a Lewis structure?
These instructions outline the Kelter strategy to draw Lewis structures for molecules.- Step 1: Find the Total Number of Valence Electrons.
- Step 2: Find the Number of Electrons Needed to Make the Atoms "Happy"
- Step 3: Determine the Number of Bonds in the Molecule.
- Step 4: Choose a Central Atom.
Which elements are exceptions to the octet rule?
The two elements that most commonly fail to complete an octet are boron and aluminum; they both readily form compounds in which they have six valence electrons, rather than the usual eight predicted by the octet rule.What are examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.



