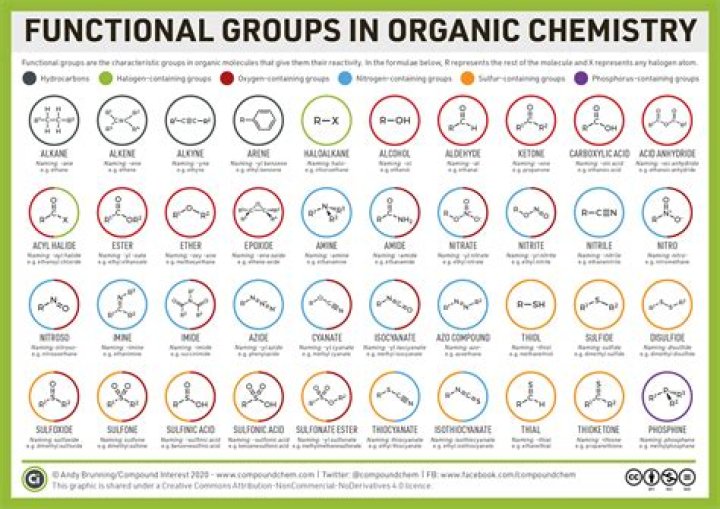
What are functional groups in molecules?
.
Considering this, what are functional groups examples?
Each type of organic molecule has its own specific typeof functional group. Functional groups in biologicalmolecules play an important role in the formation of molecules likeDNA, proteins, carbohydrates, and lipids. Functional groupsinclude: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate,and sulfhydryl.
Also, what do functional groups do? In organic chemistry , functional groups arespecific groups of atoms within molecules, that areresponsible for the characteristic chemical reactions of thosemolecules. The same functional group will undergo the sameor similar chemical reaction(s) regardless of the size of themolecule it is a part of.
Also to know is, what are the 7 functional groups?
There are 7 important functional groups inthe chemistry of life: Hydroxyl, Carbonyl, Carboxyl, Amino, Thiol,Phosphate, and aldehyde groups.
What are the characteristics of functional group?
A functional group makes up part of a largermolecule. For example, -OH, the hydroxyl group thatcharacterizes alcohols, is an oxygen with a hydrogen attached. Itcould be found on any number of different molecules. Just aselements have distinctive properties, functional groups havecharacteristic chemistries.
Related Question Answers