What affects Solid pressure?
.
Subsequently, one may also ask, what factors affect pressure in solids?
A solid resting on a horizontal surface exerts a normal contact force equals to its weight. The pressure of the solid on the surface depends on the area of contact. (b) the area of contact between the two surfaces. The greater the force or the smaller the area the greater the pressure.
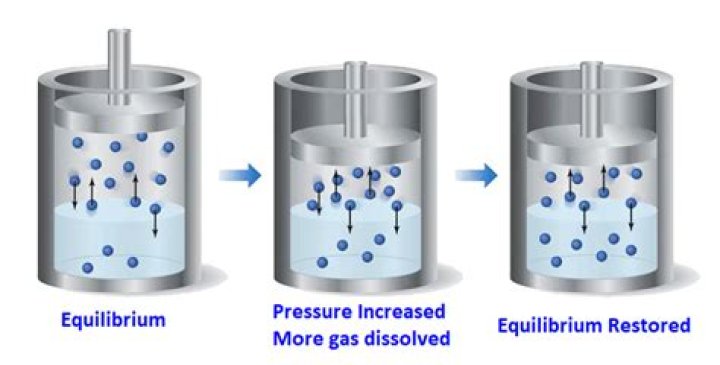
Additionally, how does pressure affect a reaction? When you increase the pressure, the molecules have less space in which they can move. That greater density of molecules increases the number of collisions. When you decrease the pressure, molecules don't hit each other as often and the rate of reaction decreases. Pressure is also related to concentration and volume.
Considering this, does Solid have pressure?
Yes, all the solids have vapour pressure. Vapour pressure can be defined as the pressure reached when a condensed phase is in equilibrium with its own vapour. For most solids this pressure is very low, but some notable examples are naphthalene, dry ice and ice.
What is the effect of pressure on state of matter?
Effect of pressure But if the pressure is changed, the temperature at which the material changes its state also changes. The higher the pressure, the higher the temperature needed to change the state of the material.
Related Question AnswersHow does a solid exert pressure?
A solid exerts pressure only on its base downwards i.e. a solid will exert pressure in the direction of its weight which is downward. while a fluid exerts pressure at all points in all directions .How do you find the pressure of something?
Pressure and force are related, and so you can calculate one if you know the other by using the physics equation, P = F/A. Because pressure is force divided by area, its meter-kilogram-second (MKS) units are newtons per square meter, or N/m2.What is solid pressure?
Pressure. Pressure is the force per unit area. This means that the pressure a solid object exerts on another solid surface is its weight in newtons divided by its area in square metres.How can we increase pressure?
Summary- An increase in the number of gas molecules in the same volume container increases pressure.
- A decrease in container volume increases gas pressure.
- An increase in temperature of a gas in a rigid container increases the pressure.
How do you increase and decrease the value of pressure?
To increase pressure - increase the force or reduce the area the force acts on. To cut up your dinner you can either press harder on your knife or use a sharper one (sharper knives have less surface area on the cutting edge of the blade). To reduce pressure - decrease the force or increase the area the force acts on.What happens when pressure increases?
PRESSURE AND TEMPERATURE RELATIONSHIP. In a closed system where volume is held constant, there is a direct relationship between Pressure and Temperature. For example, when the pressure increases then the temperature also increases. When the pressure decreases, then the temperature decreases.Is pressure a force?
Pressure. Pressure is defined as force per unit area. It is usually more convenient to use pressure rather than force to describe the influences upon fluid behavior. The standard unit for pressure is the Pascal, which is a Newton per square meter.How does temperature affect a solid?
Temperature has a direct effect on whether a substance exists as a solid, liquid or gas. Generally, increasing the temperature turns solids into liquids and liquids into gases; reducing it turns gases into liquids and liquids into solids.What happens to water when pressure increases?
When you maintain this temperature and increase the pressure in the indestructible container, water changes from a liquid phase to a solid phase when the pressure rises above 1GPa. Upon increasing the pressure further, the crystal structure of the ice will change from ice VI to ice VII and ice X to ice XI.What exactly is vapor pressure?
The vapor pressure of a liquid is the equilibrium pressure of a vapor above its liquid (or solid); that is, the pressure of the vapor resulting from evaporation of a liquid (or solid) above a sample of the liquid (or solid) in a closed container. Examples: substance. vapor pressure at 25oC.What happens to molecules when pressure increases?
With more particles there will be more collisions and so a greater pressure. Because the area of the container has increased, there will be fewer of these collisions per unit area and the pressure will decrease. Volume is inversely proportional to pressure, if the number of particles and the temperature are constant.What happens when a solid is cooled?
Students should realize that if you heat a solid, the atoms or molecules move faster and move further apart. If you cool a solid, the molecules move more slowly and move a little closer together.What do you mean by pressure?
Pressure is defined as the physical force exerted on an object. The force applied is perpendicular to the surface of objects per unit area. Unit of pressure is Pascals (Pa).Does higher vapor pressure mean faster evaporation?
The greater the pressure it exerts, the weaker the intermolecular forces between molecules in its liquid state; the more volatile the liquid; the lower the boiling point and the faster its evaporation rate. The higher the vapor pressure of a material at a given temperature, the lower the boiling point.What affects reaction rate?
Factors That Affect Reaction Rates. Factors that influence the reaction rates of chemical reactions include the concentration of reactants, temperature, the physical state of reactants and their dispersion, the solvent, and the presence of a catalyst.In what manner will increase of pressure affect the equation?
Increasing the pressure on a reaction involving reacting gases increases the rate of reaction. Changing the pressure on a reaction that involves only solids or liquids has no effect on the rate. If you increase the pressure of a gas, you squeeze it into a smaller volume.What factors affect reaction rate?
The factors that affect reaction rates are:- surface area of a solid reactant.
- concentration or pressure of a reactant.
- temperature.
- nature of the reactants.
- presence/absence of a catalyst.



