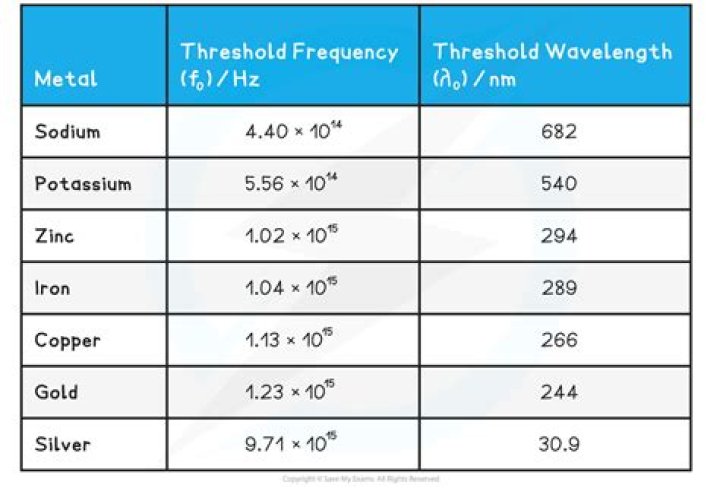
Work function is different for different metals. A photon having an energy at least equal to the work function can eject electron from the metal, frequency of such a photon whose energy is just equal to the work function is called threshold frequency..
Also, what is the difference between work function and threshold energy?
Although the work function specifically refers to the energy that needs to be put in, and the threshold energy refers to the frequency required to eject an electron, they are the same thing when calculating with the equation.
One may also ask, what is definition of threshold frequency? Threshold frequency is defined as the minimum frequency of light which causes electrons to be emitted from a metal surface. If no electrons are ejected, this means that the frequency of the light is less than the threshold frequency.
Likewise, what is the work function equal to?
In the photoelectric effect, the work function is the energy that must be supplied to overcome the attractive forces that hold an electron in the metal. For mercury, the work function is equal to 435 kJ mol-1of photons.
What is the formula for work function?
h = the Plank constant 6.63 x 10-34 J s. f = the frequency of the incident light in hertz (Hz) &phi = the work function in joules (J) Ek = the maximum kinetic energy of the emitted electrons in joules (J)
Related Question Answers
What is the symbol for work function?
The maximum kinetic energy of the electron is the energy of the photon minus the threshold energy. This threshold energy we call the "work function" and we give it the symbol Φ.What is the relation between threshold frequency and work function?
Threshold frequency is defined as the minimum frequency of incident light which can cause photo electric emission i.e. this frequency is just able to eject electrons with out giving them additional energy. Minimum amount of energy which is necessary to start photo electric emission is called Work Function.What is stopping voltage?
The stopping potential is defined as the potential necessary to stop any electron (or, in other words, to stopeven the electron with the most kinetic energy) from 'reaching the other side'. As you already stated, the maximum kinetic energy is given by.What is threshold energy in chemistry?
Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules considered as hard spheres, resulting in a chemical reaction. Therefore, Threshold energy= Average of the initial kinetic energy possessed by the reactants + Activation energy (Ea)How do you find the energy of a photon?
To figure out the energy, we use the E = hf equation. The energy of each photon is equal to Planck's constant, multiplied by the frequency of the light, h is always 6.63 * 10^-34 Joule seconds, and the frequency is 6 * 10^14 Hz. Plug those in and solve, and we get 4 * 10^-19 Joules.What is the value of threshold frequency?
Threshold frequency is the minimum frequency of light that, when incident on a material, will eject a photoelectron. To calculate this, you will need the energy of the light incident on the material and the kinetic energy of the photoelectron ejected.What is threshold wavelength?
The threshold wavelength ( more commonly threshold frequency) is the wavelength/ frequency of a photon with just sufficient energy to This probably refers to the photoelectric effect. Photons have an energy E=hf or E=hc/lambda. To release an electron from a metal surface, the photon must have sufficient energy.What is the formula of threshold wavelength?
To Find: Threshold wavelength of metal = λo =? The frequency of incident light is less than the threshold frequency. No photoelectrons will be emitted from the metal surface. Ans: The threshold frequency is 1.2 x 1015 Hz and no photoelectron will be emitted.How do you find the stopping potential of a work function?
Given: Stopping potential = Vs = 3 V, work function = Φ = 3.63 eV = 3.63 x 1.6 x 10-19 J, speed of light = c = 3 x 108 m/s, Planck's constant = h = 6.63 x 10-34 Js, Charge on electron = e = 1.6 x 10-19 C. To Find: frequency of radiation = ν =?What is the threshold frequency of sodium?
Sodium metal has a threshold frequency of 4.40×1014 Hz.What is the threshold frequency of cesium?
Cesium metal has a threshold frequency of 4.38 x 10^14 Hz.Why is there a threshold frequency?
Threshold Frequency. The threshold frequency is the frequency of light that carries enough energy to dislodge an electron from an atom. This energy is entirely consumed in the process (see References 5). Therefore, the electron gets no kinetic energy at the threshold frequency and it is not released from the atom.Why does the photoelectric effect prove light is a particle?
The photoelectric effect supports a particle theory of light in that it behaves like an elastic collision (one that conserves mechanical energy) between two particles, the photon of light and the electron of the metal. The minimum amount of energy needed to eject the electron is the binding energy, BE .What is the significance of Planck's constant?
The Planck constant (Planck's constant) links the amount of energy a photon carries with the frequency of its electromagnetic wave. It is named after the physicist Max Planck. It is an important quantity in quantum physics.What is Einstein's photoelectric equation?
: an equation in physics giving the kinetic energy of a photoelectron emitted from a metal as a result of the absorption of a radiation quantum: Ek=hν−ω where Ek is the kinetic energy of the photoelectron, h is the Planck constant, ν is the frequency associated with the radiation quantum, and ω the work function of theWhat is threshold value?
threshold value. [′thresh‚hōld ‚val·yü] (computer science) A point beyond which there is a change in the manner a program executes; in particular, an error rate above which the operating system shuts down the computer system on the assumption that a hardware failure has occurred.What is the symbol of threshold frequency?
electromagnetic phenomena The photoelectric threshold frequency, symbolized by the Greek letter nu with subscript zero, ν0, is that frequency at which the effect is barely possible; it is given by the ratio of the work function symbolized by the Greek letter psi, ψ, to Planck's constant (ν0 =…What is the stopping potential?
Definition. The stopping potential is defined as the potential necessary to stop any electron (or, in other words, to stop even the electron with the most kinetic energy) from reaching the other side. What happens in photoelectric effect is that, an incident radiation strikes the surface of the metal.