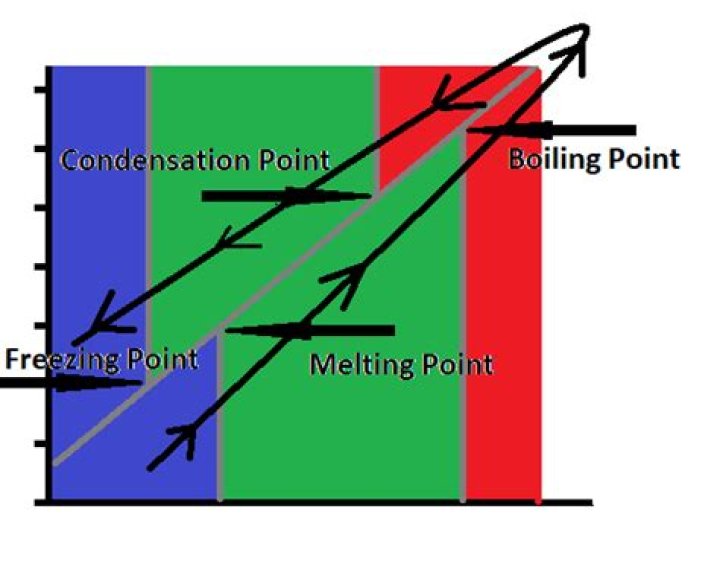
The melting point of a solid is the temperature at which the vapor pressure of the liquid phase and the solid phase are equal and at equilibrium. The freezing point of water and other liquids may be the same temperature as the melting point. It won't be higher, but it could easily be lower..
Likewise, people ask, is the freezing point the same as the melting point?
Freezing and melting are just different terms used to describe the transition between a material's solid and liquid states. Freezing usually means that energy is being removed from a material (liquid is changing to solid) and melting usually means that energy is being added to a material (solid is changing to liquid).
Also Know, what do the melting point and freezing point of a substance have in common? Common fluids and their freezing and melting points Melting point - the temperature at which a solid turns into a liquid. Freezing point - the temperature at which a liquid turns into a solid. The melting and freezing point changes with pressure, but normally they are given at 1 atm.
Similarly one may ask, are the melting point temperature of solid and freezing point of liquid same or different?
Answer: The boiling point is the temperature at which a material changes from a liquid to a gas (boils) while the melting point is the temperature at which a material changes from a solid to a liquid (melts). Keep in mind that a material's melting point is the same as its freezing point.
Why the freezing point and melting point of water is the same?
The freezing point or melting point of water is the temperature at which water changes phase from a liquid to a solid or vice versa. The freezing point describes the liquid to solid transition while the melting point is the temperature at which water goes from a solid (ice) to liquid water.
Related Question Answers
Why does water freeze?
Freezing happens when the molecules of a liquid get so cold that they slow down enough to hook onto each other, forming a solid crystal. For pure water, this happens at 32 degrees Fahrenheit, and unlike most other solids, ice expands and is actually less dense than water.Does water freeze at 0?
We've all been taught that water freezes at 32 degrees Fahrenheit, 0 degrees Celsius, 273.15 Kelvin. That's not always the case, though. Scientists have found liquid water as cold as -40 degrees F in clouds and even cooled water down to -42 degrees F in the lab.Why does water melt and freeze at 0 degrees?
Melting and Freezing As the energy in the molecules increases from a rise in temperature, the molecules start moving faster. Soon they have enough energy to break free of their rigid structure and start moving around more easily. The matter becomes a liquid. The melting point for water is 0 degrees C (32 degrees F).Does ice melt at 0 degrees Celsius?
At temperatures below 32°F (0°C), liquid water freezes; 32°F (0°C) is the freezing point of water. At temperatures above 32°F (0°C), pure water ice melts and changes state from a solid to a liquid (water); 32°F (0°C) is the melting point.Why does water not freeze at 0 degrees?
This spreading-out action of the water molecules during freezing also means that applying pressure to water lowers the freezing point. If you apply enough pressure (making it hard for the water molecules to spread out into the solid structure), you can have liquid water several degrees below zero degrees Celsius.What is I in freezing point depression?
Freezing-point depression is the decrease of the freezing point of a solvent on the addition of a non-volatile solute. Examples include salt in water, alcohol in water, or the mixing of two solids such as impurities into a finely powdered drug.Is Melting Point a physical property?
Examples of physical properties are: color, smell, freezing point, boiling point, melting point, infra-red spectrum, attraction (paramagnetic) or repulsion (diamagnetic) to magnets, opacity, viscosity and density. Note that measuring each of these properties will not alter the basic nature of the substance.Is boiling point same as condensation point?
~ The boiling point of a substance is the temperature at which it changes from a liquid to a gas. The condensation point of a substance is the temperature at which it changes from a gas to a liquid.What is the freezing point?
Freezing point is the temperature at which a liquid becomes a solid at normal atmospheric pressure. Alternatively, a melting point is the temperature at which a solid becomes a liquid at normal atmospheric pressure.What increases melting point?
Melting Point[edit] 1. As the atomic number of elements increases, the melting point increases because there are more electrons around the nucleus, which creates a stronger negatively-charged force. With stronger forces, the melting point rises.How does water affect melting point?
Water freezes at the same temperature and turns into ice. It's difficult to heat solids to temperatures above their melting points, so finding the melting point is a good way to identify a substance.Do all liquids freeze?
Things can be done to liquids to change them, but not all liquids will respond the same way. Freezing liquids is one way to change them from a liquid to a solid. Water will freeze faster than liquids with salt or sugar in them. Some liquids freeze faster than others because of viscosity, or thickness of the liquid.What are volatile liquids?
Volatile liquids are liquids which have a very high tendency to convert to the gas phase. This is dependent on the vapor pressure of the liquid. Volatile liquids have a vapor pressure higher than the normal atmospheric pressure at normal room temperature.What is the melting point in Celsius?
Celsius. Celsius or centigrade is a unit of measurement of temperature. The freezing/melting point of water is about zero degrees celsius (0 °C) at a pressure of 1 atmosphere. The boiling point of water is about on one hundred degrees celsius (100 °C) at a pressure of 1 atmosphere.Why does water have a low melting point?
Water molecules are attached to each other by hydogen bonds which is slightly stronger than van dar Waals force; therefore, it is liquid at room temperature. Still it required very low energy to eliminate the H-bond energy compared with the ionic bonds. Therefore, its melting point is comparatively lower.Does saltwater freeze?
Ocean water freezes just like freshwater, but at lower temperatures. Fresh water freezes at 32 degrees Fahrenheit but seawater freezes at about 28.4 degrees Fahrenheit , because of the salt in it. When seawater freezes, however, the ice contains very little salt because only the water part freezes.Is there any liquid that Cannot freeze?
There are no liquids that do not freeze. However, under certain conditions, it is possible to cool a liquid below its normal freezing point. Such a substance is called a supercooled liquid. Currently, no such liquid has been discovered; thus, every liquid freezes if cooled to a sufficiently low temperature.Does vinegar freeze?
Freezing of vinegar. Concentrated acetic acid freezes around seventeen degree Celsius. Water freezes at zero degree Celsius. But Vinegar has a very low freezing point , that is around minus two degree celcius.At what temperature does turpentine freeze?
Molecular weight=136–140 (approx); Specific gravity (H2O:1)=0.84–0.87 @ 20°C; Boiling point=153.8°C–170°C; Freezing/Melting point=−50°C to −60°C; Vapor pressure=4 mmHg; Flash point=35°C (cc), also listed @ 30°C–46°C (cc); Autoignition temperature=253°C, also listed @ 220°C–255°C.