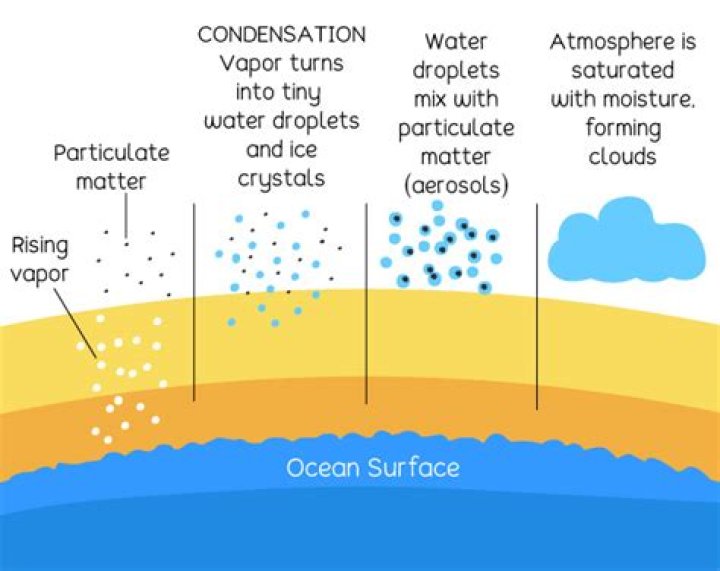
Condensation is the change of the physical state of matter from the gas phase into the liquid phase, and is the reverse of vaporization. It can also be defined as the change in the state of water vapor to liquid water when in contact with a liquid or solid surface or cloud condensation nuclei within the atmosphere..
Subsequently, one may also ask, is condensation of water vapor a physical or chemical change?
Explanation: Condensation is not a chemical change because no new compounds result, and the process can be reversed by adding energy or removing the pressure. The condensation of water vapor is the most common example because it is happening around us on a continuous basis.
Likewise, is water to gas a physical change? – In a physical change, the chemical substances DOES NOT become a new substance. – Example: water can change state from being a solid (ice) to a liquid (water) to a gas (water vapour). – Physical changes can always be reversed.
Also Know, is water vapor a physical change?
Water, ice, and water vapor are all the same basic substance. As water turns into ice or water vapor, the water molecules themselves do not change. Changes in state are physical changes because changes in state do not change the basic substance.
Is water a physical or chemical change?
Physical and Chemical Changes. A physical change is any change NOT involving a change in the substance's chemical identity. There is no effect on the chemical identity of the substance. For example, water remains water, no matter if it solid, liquid or gas.
Related Question Answers
What are three examples of physical changes?
Examples of Physical Changes - Crushing a can.
- Melting an ice cube.
- Boiling water.
- Mixing sand and water.
- Breaking a glass.
- Dissolving sugar and water.
- Shredding paper.
- Chopping wood.
Is melting a chemical or physical change?
Chemical change is when water is transformed into hydrogen gas and oxygen gas. Physical change is when you transform water into ice or steam (solid-liquid-gas). Thus, melting is a physical change.Is corrode a physical change?
Corrosion is a chemical change. Combinations of elements or compounds in a system that can combine and go into a more stable stable state (lower energy state) eventually do so. An example is a steel part rusting as the iron combines with oxygen in the air (salt and water speed this up). Corrosion is a chemical change.Is condensation a chemical reaction?
A condensation reaction is a chemical reaction between two compounds where one of the products is water, ethanol, acetic acid, hydrogen sulfide, or ammonia.Is digesting food a chemical change?
Food is chemically changed in digestion when new, smaller substances are formed. These chemical changes are examples of chemical digestion. Chemical digestion begins in the mouth when enzymes in saliva begin to break down carbohydrates. Most chemical changes in digestion occur in the small intestine.Is frying an egg a physical change?
Frying an egg is a chemical change. Any substance that changes form, produces gas bubbles or changes color has undergone a chemical change. A chemical change cannot be reversed. For example, once an egg has been fried, it cannot ever go back to its original form.Is dry ice in water a chemical change?
It's not really a chemical reaction, the water is simply turning the dry ice into a gas again. The mist that forms when water is poured onto dry ice is just that: water vapor condensing in the air from all the very cold gas being thrown off by the dry ice turning into gaseous carbon dioxide.Is milk souring a chemical change?
Souring milk is not something you can reverse, and the process of it souring produces new molecules. Some other examples of chemical changes would be things that involve burning, the creation of a new gas or bubbles, or change in color, like the formation of rust.Is burning wood a physical change?
Burning of wood is a chemical change as new substances which cannot be changed back (e.g. carbon dioxide) are formed. For example, if wood is burned in a fireplace, there is not wood anymore but ash. Compare: Physical change - The opposite of a chemical change is a physical change.Is boiling salt water a physical change?
There are many simple examples of physical changes, like melting ice (solid water) to liquid then boiling of water turning liquid to steam (gaseous water). A somewhat more interesting example is dissolving salt into water.Is soda fizzing a chemical change?
A carbonated beverage container is opened, producing a fizzing sound and carbon dioxide (CO ) bubbles. This would be a chemical change because when you open the soda you are releasing the bubbles therefore releasing the gas within the bubbles. This is considered a physical change there is no new substance being formed.Which is a chemical property?
A chemical property is any of a material's properties that becomes evident during, or after, a chemical reaction; that is, any quality that can be established only by changing a substance's chemical identity. They can also be useful to identify an unknown substance or to separate or purify it from other substances.Is condensation a physical property?
Answer and Explanation: Condensation is a physical change. In condensation, a gas turns into a liquid. The molecules of the gas do not change when they turn into the liquid,Is cooking an egg a chemical change?
Cooking an egg is a set of chemical changes because chemical bonds are broken and new ones are made, resulting in the formation of new substances. In this case many of the bonds broken are those which cause the egg's proteins to be folded into a globular shape.Which description of salt is a physical property?
The physical properties of the salt determine how the salt is produced and used. Salt is a crystalline solid and found in the cubic form along with the seawater as the salty lake water evaporates due to heat energy of sun. It is isomeric in nature. The structure of salt is orthorhombic and white in color.What are examples of physical properties?
physical and chemical properties. Examples of physical properties are: color, smell, freezing point, boiling point, melting point, infra-red spectrum, attraction (paramagnetic) or repulsion (diamagnetic) to magnets, opacity, viscosity and density. There are many more examples.How can you cause a physical change?
When you melt an ice cube (H2O), you have a physical change because you add energy. You added enough energy to create a phase change from solid to liquid. Physical actions, such as changing temperature or pressure, can cause physical changes. No chemical changes took place when you melted the ice.What happens during melting?
Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that the motion overcomes the attractions so that the molecules can move past each other as a liquid.Is baking a cake a physical change?
The chemical change occurs when the heat activates the leavener in the cake and cause it to rise. The physical change occurs when the cake turns from a liquid batter to a solid cake structure.