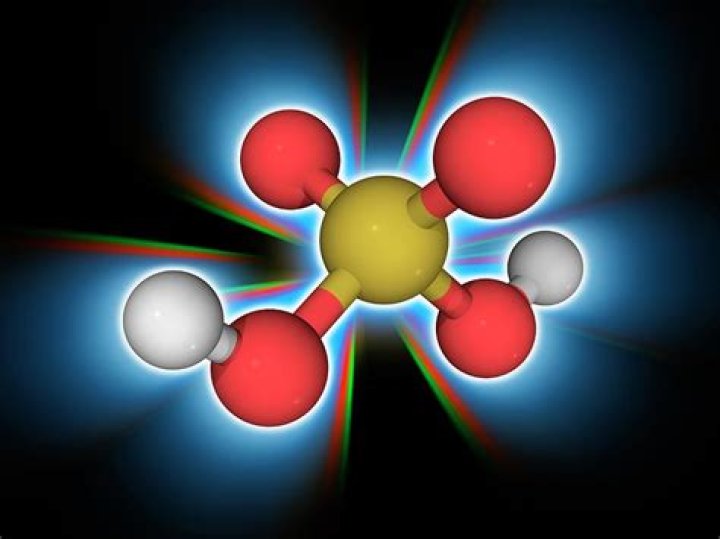
Sulfuric Acid, H2SO4 is a chemical compound made up of two hydrogen atom, one sulfer atom, and four oxygen atoms. Sulfuric acid is a strong acid, soluble in water, very polar and is an excellent solvent..
Accordingly, is sulfuric acid a mixture?
because you used the term “concentrated sulfuric acid” it is necessarily a mixture (the key term being “concentrated”). It IS possible to create 100% H2SO4. Whether that is considered a 'pure substance' or a mixture of H2O+SO3 or a mixture of H2, S, and O2 depends on the conditions of your observation.
what is the chemical composition of sulfuric acid? H2SO4
Keeping this in consideration, is Sulphuric acid a element or compound?
Sulfuric acid
What is the state of sulfuric acid?
Sulfuric acid is a colorless oily liquid. It is soluble in water with release of heat. It is corrosive to metals and tissue. Sulfuric acid, spent appears as a black oily liquid.
Related Question Answers
What is the world's strongest acid?
The carborane superacids may be considered the world's strongest solo acid, as fluoroantimonic acid is actually a mixture of hydrofluoric acid and antimony pentafluoride.Where is sulfuric acid in nature?
Sulfuric acid is found in battery acid and in Earth's acid rain.What is the PH of Sulphuric acid?
Originally Answered: What is the pH value of sulphuric acid? The Ph is around 2. It could be 1 if it is even more concentrated. Only super concentrated H2SO4 would have a Ph value of 7, because it doesn't have water & therefore cannot really be called an acid.Is milk a mixture?
Milk is a mixture of liquid butterfat globules dispersed and suspended in water. Colloids are generally considered heterogeneous mixtures, but have some qualities of homogeneous mixtures as well.Why is Sulphuric acid called the king of chemicals?
It is called king of acid because of its direct and indirect applications in manufacture of many chemicals including fertilisers. Sulphuric acid is used to clean up rust from steel rolls and soap. It is ideal to call sulphuric acid as king of chemicals . It is corrosive acts as good dehydrant.Does Sulphuric acid evaporate?
The equilibrium, saturated vapour pressure of sulphuric acid is exceedingly small at ambient temperatures. The bp of very concentrated (fuming at >98%) acid is 337 C. Significant evaporation only occurs at temperatures in excess of 150 C and at high acid concentrations (see graph).How strong is sulfuric acid?
Sulfuric acid, H2SO4, is a strong mineral acid with a melting point of 10*C and a boiling point of 290*C. Sulfuric acid reacts with copper oxide to form copper sulfate. Sulfuric acid, H2SO4, is a clear, colorless, odorless, and highly corrosive liquid.Is NaOH an acid or base?
NaOH is a base because when dissolved in water it dissociates into Na+ and OH- ions. It is the OH- (hydroxyl ion) which makes NaOH a base. In classical term a base is defined as a compound which reacts with an acid to form salt and water as depicted by the following equation. NaOH+HCl=NaCl+H2O.What is h2 called?
H2 is also called molecular hydrogen.It consists of two protons and two electrons. Consequently it is the most common form of Hydrogen because it is stable with a neutral charge. H2 is not a free radical. It is the antioxidant in 'hydrogen-rich' water. H2 is the smallest molecule in the universe.Will sulfuric acid dissolve rubber?
Acids. Acids are chemical compounds with a pH less than 7.0. Hydrochloric acid, hydrofluoric acid and sulfuric acid attack and degrade Buna-N rubber. The preservative benzoic acid is detrimental to EPDM rubber seals.How Sulphuric acid is formed?
Sulfuric acid is produced from sulfur, oxygen and water via the contact process. In the first step, sulfur is burned to produce sulfur dioxide. This is then oxidised to sulfur trioxide using oxygen in the presence of a vanadium(V) oxide catalyst.How many elements are in sulfuric acid?
Sulfuric Acid, H2SO4 is a chemical compound made up of two hydrogen atom, one sulfer atom, and four oxygen atoms.Can you buy sulfuric acid?
Battery acid, which may be purchased at an automotive supply store, is approximately 35% sulfuric acid. In many cases, this will be strong enough for your activities, but if you need concentrated sulfuric acid, you just need to remove the water.What products contain sulfuric acid?
Household Products Containing Sulfuric Acid This makes it an excellent choice for products such as toilet bowl cleaners and drain cleaners/openers. It may also be used in powdered laundry detergents, hand soap, dishwashing liquid and pet products.How dangerous is sulfuric acid?
Sulfuric acid is a highly corrosive chemical that is potentially explosive in concentrated form. It can cause severe skin burns, can irritate the nose and throat and cause difficulties breathing if inhaled, can burn the eyes and possibly cause blindness, and can burn holes in the stomach if swallowed.Is an orange a compound?
A Orange juice contains particles of solid (pulp) as well as liquid; it is not chemically pure. B Because its composition is not uniform throughout, orange juice is a heterogeneous mixture. A Aluminum oxide is a single, chemically pure compound. A Selenium is one of the known elements.Is sulfuric acid flammable?
Concentrated Sulfuric Acid will react with many organic materials and may cause fire due to the heat of the reaction. Not flammable, but reacts with most metals to form explosive/flammable hydrogen gas.What are the properties of sulfuric acid?
Physical properties: H2SO4 is a colorless or slightly yellow viscous liquid with a pungent odor. It has a density of 1.84 g/mL, boiling point of 337 °C, and melting point of 10 °C. "Concentrated" sulfuric acid is 98% in water, and is the most stable form.Why is sulfuric acid important?
This is the substance sold as concentrated sulfuric acid. Because of this high affinity for water, sulfuric acid is often used as a drying agent in the production of explosives, dyes, detergents, and various anhydrous (water-free) materials.