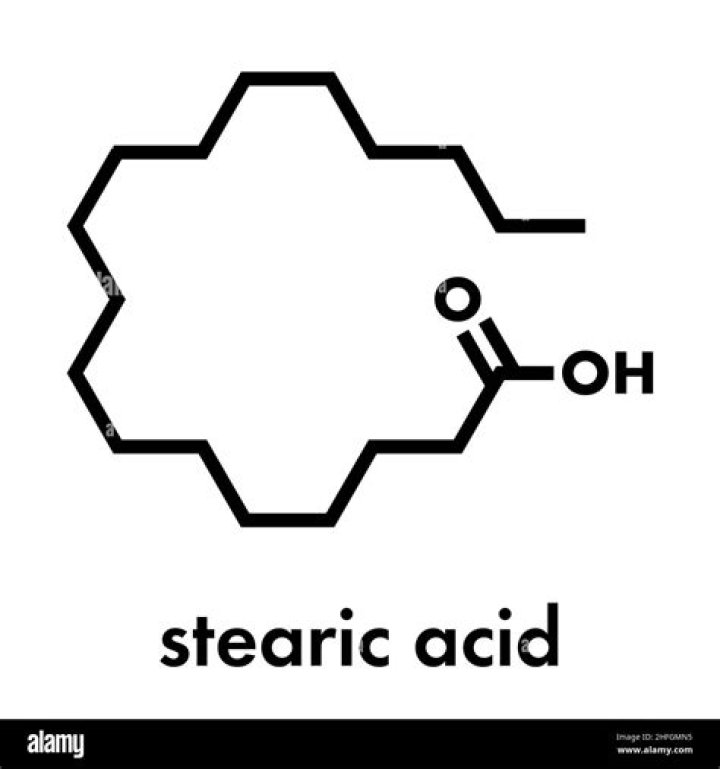
Stearic acid is a saturated fatty acidthat can deposit on the surface in special conditions. Thisacid is insoluble in water and soluble inethanol..
Regarding this, why is stearic acid insoluble in water?
The tail is basically an alkane, and so readilydissolves in fat, oil and grease, but not in water.Thus the tail is said to be hydrophobic (water-hating). Thehead-group however is polar, and so easily dissolves inwater (hydrophilic - water-loving) and will notdissolve in oil or grease.
Also Know, is stearic acid conductive in water? Stearic acid is obtained from fats and oils bythe saponification of the triglycerides using hot water(about 100 °C). Fats and oils rich in stearic acid aremore abundant in animal fat (up to 30%) than in vegetable fat(typically <5%).
Correspondingly, is stearic acid water or oil soluble?
Stearic Acid is a waxy, colorless or white solidthat exudes a mild odor. It is soluble in oil butonly slightly dissolves in water, thus it floats.Stearic Acid is a long-chain fatty acid that, due toits 18-carbon chain, is also referred to as OctadecanoicAcid.
What does stearic acid smell like?
Pure stearic acid is a white, waxy solidcrystalline material that melts at 156°F (69°C). It isodorless and tasteless. These trace impurities can cause theacid to vary in molecular weight, solubility, melting point,color, odor, and other physical and chemicalproperties.
Related Question Answers
What dissolves stearic acid?
Stearic acid is soluble in organic solvents suchas ethanol, DMSO, and dimethyl formamide. The solubility ofstearic acid in these solvents is approximately 20, 10, and30 mg/ml, respectively. Description. Stearic acid is along-chain saturated fatty acid.What is stearic acid in skin care?
Fatty acid used as an emollient and emulsifier.It has been shown to protect skin's surface against waterloss and help shore up skin's protective barrier. Stearicacid may be synthetic or animal-derived.What does stearic acid do in lotion?
For creams and lotions, it can be usedanywhere from 2-30%. Stearic Acid: This cost effectiveco-emulsifier is often used to thicken lotion and otheremulsified products. Stearic acid is derived fromvegetables, and can also be used to harden soaps at a rate of .5%of your oils.Is stearic acid covalent?
Also, because it is under the "umbrella" of acovalent bond, we believe it will have a fast melting time.Non-polar covalent compounds (stearic acid andsalicylic acid) are all insoluble in water, soluble intoluene, have low melting times, and are poor conductors ofelectricity because there are no ions.What is PH of stearic acid?
mixture of fatty acids, primarily stearicand palmitic acid (acc. to Ph Helv)Is stearic acid soluble in NaOH?
All carboxylic acids are both aliphatic andaromatic so they soluble in water. NaOH isn't aliquid, it's a solid which can be dissolved in water to forma solution. The solubility of stearic acid with H Oand NaOH is match with the theory but the solubilty ofstearic acid with ether does not match with thetheory.What products contain stearic acid?
a: Specific foods contributing at least 1% ofstearic acid in descending order: Nuts/seeds and nut/seedmixed dishes, pork and pork mixed dishes, potato/corn/other chips,quickbreads, salad dressing, soups, crackers, other whitepotatoes.What foods contain stearic acid?
Stearic acid: The top five sources of stearicacid are grain-based desserts; regular cheese; sausage, franks,bacon, and ribs; chicken and chicken mixed dishes; andpizza.Is stearic acid an emulsifier?
No, stearic acid is not an emulsifier. Itcan thicken a product, but that's all it can do.What does stearic acid do in soap?
By lowering the surface tension of oil, stearicacid allows water to combine with the oil molecules and washthem away. As a result, stearic acid helps to remove dirt,sweat and excess sebum from the skin and hair. This makes it auseful ingredient in cleansers, body washes and shampoos as well asbar soaps.What is vegetable stearic acid made from?
Stearic acid, also called OctadecanoicAcid, one of the most common long-chain fatty acids,found in combined form in natural animal and vegetable fats.Commercial “stearic acid” is a mixture ofapproximately equal amounts of stearic and palmiticacids and small amounts of oleic acid.How dangerous is stearic acid?
HUMAN STUDIES: The greatest danger from ingestionof large quantities of stearic acid is intestinalobstruction. Skin sensitization is unusual. Aspiration orinhalation of stearic acid could cause chemical pneumonitis.Implantation of stearic acid will cause foreign bodyreaction.What is freezing point of stearic acid?
The exact melting and freezing points of thestearic acid may not be exactly the same and will depend onthe purity of the product and where it was purchased from, but areusually around 55-70 ˚C.Is sucrose conductive in water?
All of the bonds in the sucrose molecule arestrong covalent bonds.Therefore there are no charged particlespresent to conduct electricity either in the solid state or insolution. Substances like sucrose which do not conductelectricity in aqueous solution are callednon-electrolytes.What type of lipid is stearic acid?
Stearic Acid is a saturated long-chain fattyacid with an 18-carbon backbone. Stearic acid isfound in various animal and plant fats, and is a major component ofcocoa butter and shea butter. Octadecanoic acid is a C18straight-chain saturated fatty acid component of many animaland vegetable lipids.Is stearic acid a surfactant?
Stearic acid is one of many fatty acidsthat occur naturally in various plants and animal derivatives. It'sfound in such products as animal tallow, cocoa butter and vegetablefats. As a surfactant, stearic acid is a powerfulcleanser [source: Personal Care Products Council].Why is stearic acid volatile?
Stearic acid is more volatile thansucrose. Stearic acid is non polar, which means it dissolvesin non polar solvent such as hexane. It is possible for a compoundto be volatile but odorless.Is stearic acid natural?
Stearic acid is a white, waxy, naturalacid found in animal and vegetable fats. Our Stearicacid is from vegetable origins (palm) and is manufactured anddesigned to blend well with your cosmetic recipes. Hydrogenation isa process in which liquid vegetable oils are converted to solid orsemi-solid fats.