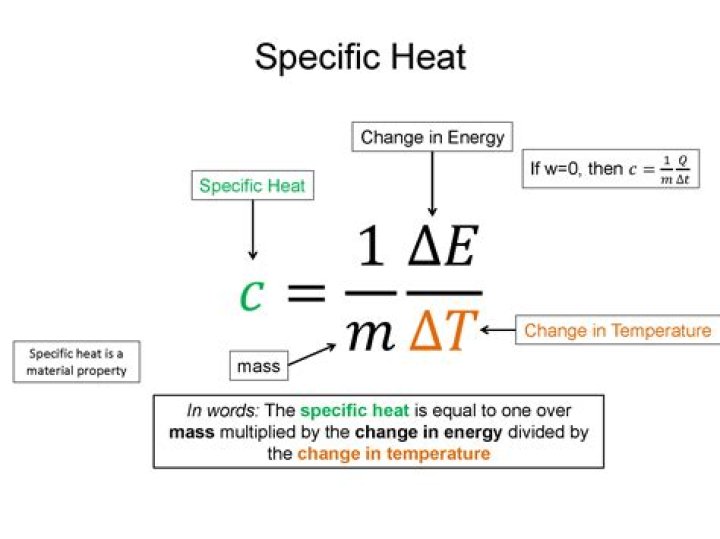
Specific heat is a characteristic property that measures how much energy it takes to raise the temperature of 1 gram of a material by 1 oC. The higher the c value is, the more difficult it is to warm up that substance..
In this way, can specific heat be used to identify a substance?
Specific heat is an intensive property (like density, color, etc.) that does not depend on the amount of a substance present. This allows substances to be identified using their specific heat. Imagine an unknown metal of known mass is heated to a known temperature.
Subsequently, question is, what does the specific heat of a substance represent? specific heat: The ratio of the amount of heat needed to raise the temperature of a unit mass of substance by a unit degree to the amount of heat needed to raise that of the same mass of water by the same amount.
Also question is, what are characteristics of substances with high specific heat and low specific heat?
Specific heat is Jg−oK . So, a high value means that it takes MORE energy to raise (or lower) its temperature. A low value means that it does not take very much energy to heat or cool it.
Why specific heat is different for different substances?
Specific heat, which is also known as thermal capacity, is the amount of heat required to raise the temperature of one gram of substance by 1°. Therefore, because mass is the only variable, so because substances have different masses, they will have different specific heats.
Related Question Answers
How can specific heat be measured?
Specific heat capacity is measured by determining how much heat energy is needed to raise one gram of a substance one degree Celsius. The specific heat capacity of water is 4.2 joules per gram per degree Celsius or 1 calorie per gram per degree Celsius.How can you identify an unknown metal?
You can identify an unknown substance by measuring its density and comparing your result to a list of known densities. Density = mass/volume. Assume that you have to identify an unknown metal. You can determine the mass of the metal on a scale.Can specific heat capacity be negative?
If the system loses energy, for example, by radiating energy into space, the average kinetic energy actually increases. If a temperature is defined by the average kinetic energy, then the system therefore can be said to have a negative heat capacity. A more extreme version of this occurs with black holes.How do scientists use specific heat?
It is basically measured by connecting a mechanical system to a calorimeter and measuring how much mechanical energy it takes to heat water by a certain temperature. Well if you add X amount of energy to a substance and it heats up by Y degrees you have its specific heat. The units of specific heat are J/(kg*K).Do metals have high or low specific heat?
It is the amount of energy (in joules) needed to raise the temperature of 1 gram of the substance by 1 °C. Specific heat is a property that is specific to a given type of matter, and substances vary in their specific heat. Metals tend to have low specific heat. Water has very high specific heat.What is specific heat used for?
Specific heat is a measure that quantifies the amount of heat required to increase the temerature of a mass of a substance. This is usually measured in Jules of heat energy needed to raise one gram of a substance by 1 deegree Celcius.Why does the specific heat change with temperature?
Specific heat is a measure of the ability of the substance to absorb heat. As the substance heats up, the average kinetic energy of the molecules increases. The collisions impart enough energy to allow rotation to occur. Rotation then contributes to the internal energy and raises the specific heat.How do we use specific heat in everyday life?
Specific heat gives the amount of heat energy required for heating a substance of 1 unit mass by 1 °C. So more the specific heat, more the energy required for heating the substance. 1) Utensils used for making tea or coffee, or cooking vegetables or rice are made of materials of low specific heat.What is an example of specific heat?
The specific heat capacity of a substance is the amount of energy required to raise the temperature of 1 kg of the substance by 1°C. Examples: 1. Calculate the energy required to increase the temperature of 2kg of water from 20°C to 100°C.What material has the highest specific heat?
kg. K−1. The specific heat often varies with temperature, and is different for each state of matter. Liquid water has one of the highest specific heats among common substances, about 4182 J/K/kg at 20 °C; but that of ice just below 0 °C is only 2093 J/K/kg.What has the highest specific heat?
Water
Why is specific heat capacity important?
Specific heat capacity is a measure of the amount of heat energy required to change the temperature of 1 kg of a material by 1 K. Hence it is important as it will give an indication of how much energy will be required to heat or cool an object of a given mass by a given amount.What is high specific heat of water?
Specific heat is the amount of energy (in Joules) that is needed to raise the temperature of a substance by one degree Celsius. Water has the highest specific heat of any other liquid. It requires 4.184 Joules to raise the temperature of one gram of water by one degree Celsius.What is meant by specific heat?
The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius. The relationship between heat and temperature change is usually expressed in the form shown below where c is the specific heat. As a result, water plays a very important role in temperature regulation.What is high specific heat?
Specific heat is defined by the amount of heat needed to raise the temperature of 1 gram of a substance 1 degree Celsius (°C). Water has a high specific heat capacity which we'll refer to as simply "heat capacity", meaning it takes more energy to increase the temperature of water compared to other substances.What is temperature a measure of?
Heat and temperature are related, but not the same. Temperature is a measure of the average kinetic energy of the particles of a substance. The higher the temperature of an object, the higher is its kinetic energy.Does specific heat change with temperature?
As the substance heats up, the average temperature of the molecules increases, so when they collide, they are more likely to impart enough energy to allow rotation and vibration to occur as the energy jumps to a higher state. With an increase in temperature the specific heat of water first decreases then increases.What is the SI unit of specific heat?
joules
What is the unit of specific heat?
Joule per kelvin