Is solubility a chemical reaction?
.
People also ask, is solubility a chemical or physical property?
Solubility is a chemical property referring to the ability for a given substance, the solute, to dissolve in a solvent. It is measured in terms of the maximum amount of solute dissolved in a solvent at equilibrium. Certain substances are soluble in all proportions with a given solvent, such as ethanol in water.
Likewise, how is solubility defined? Amount of a substance (called the solute) that dissolves in a unit volume of a liquid substance (called the solvent) to form a saturated solution under specified conditions of temperature and pressure. Solubility is expressed usually as moles of solute per 100 grams of solvent.
In this regard, is dissolving a chemical reaction?
Why Dissolving Salt Is a Chemical Change Therefore, dissolving salt in water is a chemical change. In contrast, dissolving a covalent compound like sugar does not result in a chemical reaction. When sugar is dissolved, the molecules disperse throughout the water, but they do not change their chemical identity.
Is boiling point a chemical property?
Boiling water: Boiling water is an example of a physical change and not a chemical change because the water vapor still has the same molecular structure as liquid water (H2O). If the bubbles were caused by the decomposition of a molecule into a gas (such as H2O →H2 and O2), then boiling would be a chemical change.
Related Question AnswersWhy is solubility a chemical property?
Solubility is a physical property. The reason is because it can be determined by simple observation and does not change the chemical composition of the material. If the material is soluble, the solubility is the maximum amount of material that dissolves in a solvent at a given temperature.Why is pH a chemical property?
Acidity is a chemical property because it is a measure of the presence of a particular chemical, the H+ ion (hydrogen with a positive charge. Water, H2O, is a combination of H+ and OH-, and is neutral, both electrically, and in pH, the measure of acidity. The pH will be less than 7, and the solution will be acid.What are 4 examples of chemical properties?
Examples of chemical properties include flammability, toxicity, chemical stability, and heat of combustion. Chemical properties are used to establish chemical classifications, which are used in labels on containers and storage areas.What affects solubility?
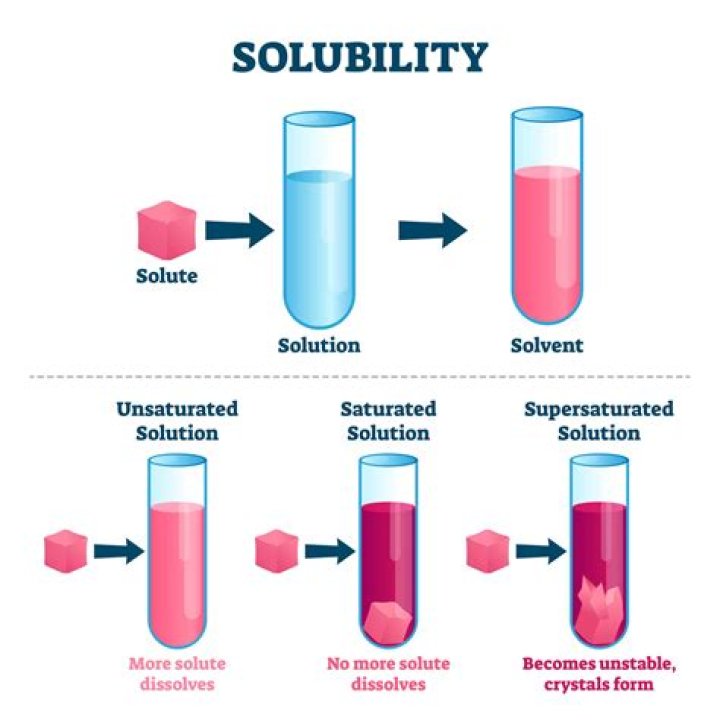
Solubility is the maximum amount of a substance that will dissolve in a given amount of solvent at a specific temperature. There are two direct factors that affect solubility: temperature and pressure. Temperature affects the solubility of both solids and gases, but pressure only affects the solubility of gases.Why is solubility important?
Solubility indicates the upper concentration a compound reaches in a solution. Solubility is a very important property in drug discovery and development, because concentration affects so many aspects of pharmacology (e.g., structure-activity relationships, efficacy, pharmacokinetics, toxicity).What is a chemical property of paper?
Introduction. A paper based product typically contains 90–99% cellulose fibers which are the primary structural element and the most important component influencing end use properties. A network of self-bonding cellulose fibers within network structure affects chemical and physical characteristics of the paper productsWhich is a chemical property?
A chemical property is a characteristic of a substance that may be observed when it participates in a chemical reaction. Examples of chemical properties include flammability, toxicity, chemical stability, and heat of combustion.Is dissolving a chemical property?
Why Dissolving Salt Is a Chemical Change Therefore, dissolving salt in water is a chemical change. The reactant (sodium chloride, or NaCl) is different from the products (sodium cation and chlorine anion). In contrast, dissolving a covalent compound like sugar does not result in a chemical reaction.Which process is a chemical reaction?
A chemical reaction is a process in which one or more substances, also called reactants, are converted to one or more different substances, known as products. A chemical reaction rearranges the constituent atoms of the reactants to create different substances as products.What is dissolving in chemistry?
In chemistry, to dissolve is to cause a solute to pass into a solution. Dissolving is also called dissolution. If dissolution is favored, the substance is said to be soluble in that solvent. In contrast, if very little solute dissolves, it is said to be insoluble.What is the science behind dissolving?
A solution is made when one substance called the solute "dissolves" into another substance called the solvent. Dissolving is when the solute breaks up from a larger crystal of molecules into much smaller groups or individual molecules. This break up is caused by coming into contact with the solvent.Is salt and ice a chemical reaction?
Salt doesn't melt ice. That's right, there is absolutely no melting going on. Instead, salt creates a chemical reaction inside ice.What is chemical method?
The chemical method refers to metal extraction by acidic or alkaline reagents (Lee and Mishra, 2018). An assortment of chemical reagents can obtain an ideal leaching that results in the extraction of heavy metals from E-waste. However, in the process, secondary pollution is generated.Is burning a chemical or physical change?
Burning of wood is a chemical change as new substances which cannot be changed back (e.g. carbon dioxide) are formed. For example, if wood is burned in a fireplace, there is not wood anymore but ash. Compare: Physical change - The opposite of a chemical change is a physical change.Is digesting food a chemical or physical change?
Chemical digestion is considered a chemical change because enzymes in the stomach and intestines break down large macromolecules into simpler molecules so that the body can more easily absorb the food.Is Rusting a chemical or physical change?
Rusting is a chemical change because the iron is changed into a new substance. Rusting is a chemical change because you start with iron and end up with iron oxide, two different substances.What are the 4 factors affecting solubility?
Factors affecting solubility- Temperature. Basically, solubility increases with temperature.
- Polarity. In most cases solutes dissolve in solvents that have a similar polarity.
- Pressure. Solid and liquid solutes.
- Molecular size.
- Stirring increases the speed of dissolving.



