Is Potassium a ionic?
.
People also ask, is potassium ionic or covalent?
Explanation: Potassium iodide (KI) forms an ionic bond. Potassium and iodine have very different electronegativities. The two atoms would form an ionic bond since ionic bonds form between atoms with a large difference in electronegativity (difference>1.7 using the Pauling scale will result in an ionic bond).
Also, is potassium and calcium an ionic compound? Potassium forms ions with a positive charge. A compound is formed when potassium reacts with chlorine, which forms ions with a negative charge. The formula of this compound would therefore be KCl. This is because calcium is in group two and so forms ions with a two positive charge.
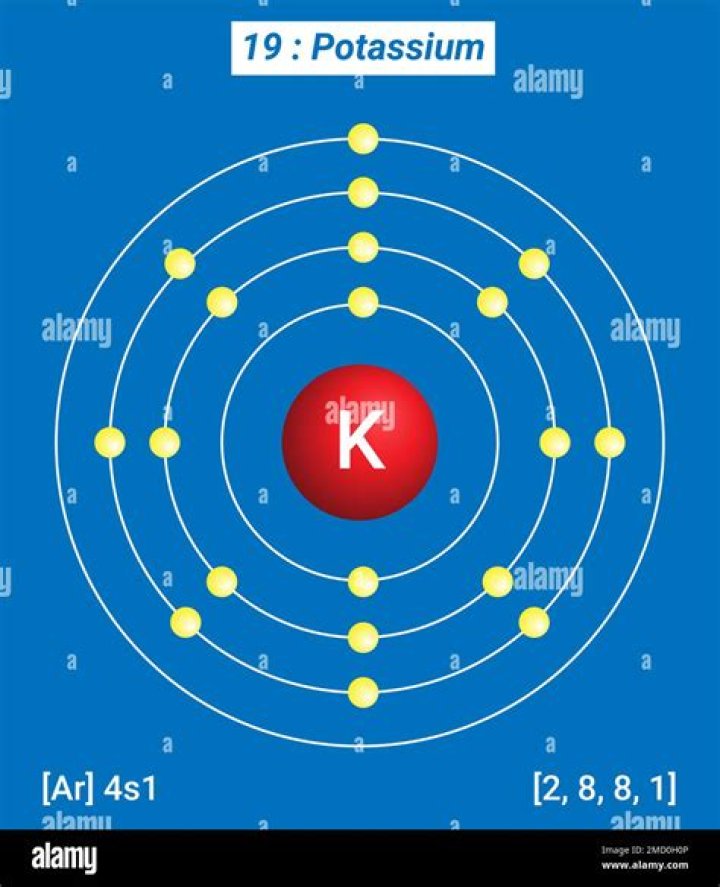
Besides, what type of ion is potassium?
Physiologically, it exists as an ion in the body. Potassium (K+) is a positively charged electrolyte, cation, which is present throughout the body in both intracellular and extracellular fluids.
Is KCl polar or nonpolar or ionic?
KCl is ionic because it contains K+ and Cl- ions in its structure.
Related Question AnswersIs HCl an ionic bond?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.Is KCl an ionic bond?
Chemical bonding is the process that links elements together to form compounds. KCl is an example of ionic bonding. Below is a simple diagram of potassium and chlorine atoms. With this electron transfer, the ionic bond in KCl is formed.What type of bond is NaCl?
Ionic compoundsIs sucrose made of ions?
Sucrose is a covalent compound. Whether a compound is ionic or covalent depends on the relative attraction the compound's atoms have for electrons. When sugar dissolves in water, the sucrose molecules remain whole, but when salt dissolves, it dissociates into individual sodium and chloride ions.Is vinegar ionic or molecular?
Its simple formula is, therefore, H2O, that of the basic solute of common household vinegar (acetic acid) is C2H4O2 (This is often written as CH3COOH to better reflect its structure), and that of common salt is NaCl. One usually describes an ionic compound only by a simple formula, though exceptions do exist.Is HCl a covalent bond?
HCl is a covalent compound. Because, both the atoms share the electrons. Moreover, HCl has intemolecular London dispersion forces, which arises to the EN (electronegativity) difference between Chlorine and Hydrogen atom. BUT: No compound is 100% ionic or covalent.Why is HCl not ionic?
Answer and Explanation: HCl is not an ionic compound it is a covalent bond. This is because chlorine and hydrogen share an electron in HCl making it a covalent bond.Is co2 a covalent bond?
Carbon dioxide is covalent bond. Carbon cannot form ionic bond because in the simplest sense carbon can neither lose 4 electrons for stablisation (Due to it's strong attraction to the nucleus) nor gain 4 electrons (Since it's small nucleus with 6 protons cannot manage extra 4 electrons).Is oxygen an ion?
Oxygen, O. Oxygen is in Group 6. It has six electrons in its outer shell. It gains two electrons from one or two other atoms in reactions, forming an oxide ion, O 2-.Is potassium ion positive or negative?
Chemicals in the body are "electrically-charged" -- when they have an electrical charge, they are called ions. The important ions in the nervous system are sodium and potassium (both have 1 positive charge, +), calcium (has 2 positive charges, ++) and chloride (has a negative charge, -).What is the best type of potassium?
There are many forms of potassium available in supplements: potassium gluconate, aspartate, chelate, chloride, phosphate, bicarbonate, citrate, and orotate. Potassium chloride is the most common type used to treat deficiency. The recommended daily intake (RDI) for potassium is 4,700 mg.What influences potassium ion?
Aldosterone binds to mineralocorticoid receptors in the cells in the cortical collecting tubule to activate and increase the number of sodium channels at the luminal membrane. Distal sodium delivery and the rate of distal tubular fluid flow are also important factors that determine the rate of potassium secretion.Is oxygen ion positive or negative?
A good example is an oxygen atom that is sharing one electron with each of two nearby hydrogen atoms, making a molecule of water H2O. An oxygen atom that has successfully donated one or two electrons or is sharing one or two electrons has lost part of its negative charge, so it is left positively charged.Why is potassium good for you?
Potassium is one of the most important minerals in the body. It helps regulate fluid balance, muscle contractions and nerve signals. What's more, a high-potassium diet may help reduce blood pressure and water retention, protect against stroke and prevent osteoporosis and kidney stones.How does potassium affect the heart?
Potassium plays a role in every heartbeat. A hundred thousand times a day, it helps trigger your heart to squeeze blood through your body. It also helps your muscles to move, your nerves to work, and your kidneys to filter blood.Is potassium an isotope?
Potassium: isotope data. Although Potassium only has two stable isotopes (K-39 and K-41), the long-lived radioisotope K-40 is often also regarded as a stable isotope. Potassium isotopes, mainly K-40 and K-41, are used to study the impact of potassium on the growth of plants and of the human cardiovascular system.How big is a potassium ion?
Intracellular K+ concentration (K+) is normally 150 mM, while the extracellular concentration is only 3.5 to 5.0 mM.What are some examples of ionic bonds?
Ionic bond examples include:- LiF - Lithium Fluoride.
- LiCl - Lithium Chloride.
- LiBr - Lithium Bromide.
- LiI - Lithium Iodide.
- NaF - Sodium Fluoride.
- NaCl - Sodium Chloride.
- NaBr - Sodium Bromide.
- NaI - Sodium Iodide.