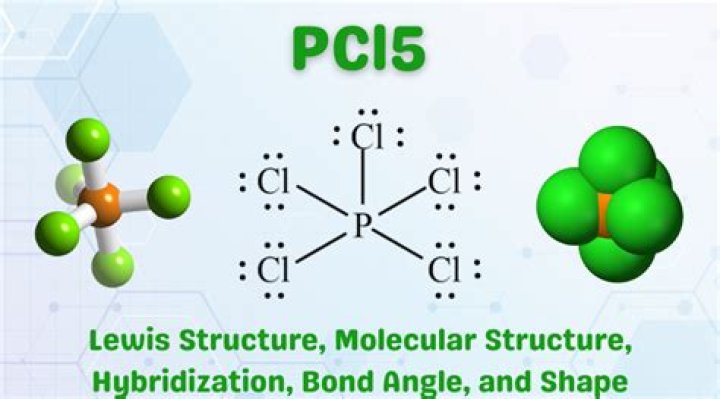
Actually PCl5 is a covalent compound. But here the P atom is sp3d hybridised and it is an non equivalent hybridization . So all the 5 P-Cl bonds are not equal in length..
Also, is PCl5 covalent or ionic?
are covalent compounds. They are not ionic. P has 5 electrons in the outer shell. Sharing 3 more electrons will make it 8 and stable (inert gas electronic configuration).
Secondly, why is PCl5 covalent? PCl5 is a non-polar covalent molecule because individual dipole moments of P-Cl bond cancel out each other but PCl3 is a polar covalent molecule. Therefore, PCl5 is more covalent than PCl3.
In respect to this, what type of compound is PCl5?
Phosphorus pentachloride
Why solid PCl5 is an ionic compound?
Lattice energy is usually the largest factor in determining the stability of an ionic solid. The extra energy gained by the lattice energy more than compensates for the energy needed to transfer a chloride ion from one PCl5 molecule to another. Thus, PCl5 exists as an ionic solid.
Related Question Answers
Is co2 a covalent bond?
Carbon dioxide is covalent bond. Carbon cannot form ionic bond because in the simplest sense carbon can neither lose 4 electrons for stablisation (Due to it's strong attraction to the nucleus) nor gain 4 electrons (Since it's small nucleus with 6 protons cannot manage extra 4 electrons).Is no2 ionic or covalent?
Answer and Explanation: NO2 or nitrogen dioxide is a covalent compound. NO2 is comprised of Nitrogen and oxygen which are both highly electronegative chemical elements.Is scl2 ionic or covalent?
The EN value of S is 2.58 and that for Cl is 3.16. The difference is 0.55 so EACH S-Cl BOND is slightly polar, but not enough to be ionic. It is covalent (molecular). Now, to determine if the molecule is polar, you need to look at the Lewis dot structure.Is pcl3 covalent or ionic?
In CCl4, atoms chlorine and carbon share electron pairs to form a covalent bond. Ionic bonds are formed by electrostatic interactions between an electropositive and electronegative atom.Is co2 an ionic compound?
Answer and Explanation: CO2 is a molecular compound. Ionic compounds are composed of a non-metal and a metal element.Is MgCl2 ionic or covalent?
Answer and Explanation: MgCl2 is an ionic compound. Magnesium is a metal with a positive charge of +2.Is PCl5 an ionic compound?
Phosphorus(V) chloride Phosphorus pentachlorideIs Na3N ionic?
Na3N has ionic bond. Na has 1 electron whereas N has 5 electrons in its outermost shell. Three sodium atoms gives up the last shell electron in order to form three Na+ anions. Thus Na3N is an ionic compound.Why PCl5 is unstable?
Answer: PCl5 is unstable because phosphorus forms 5 bonds with cl atoms out of which the two axial bonds have more Bond length than three equatorial Bond length this result in repulsion and thereby making the axial bonds weak.Is PCl5 polar or nonpolar?
The chemical compound phosphorous pentachloride, which has the chemical formula PCl5, is a non-polar molecule. The molecular geometry of phosphorous pentachloride is symmetrical, which neutralizes the bond dipoles of the molecule to make it non-polar.Is table salt a compound?
Sodium chloride
Is pcl3 soluble in water?
Water
What is a PCl5?
Hewlett-Packard developed the Printer Command Language, or PCL, for its ink-jet printers in the 1980s. PCL5 is the last version to be based on the traditional code that the computer driver sends to the printer to give it the instructions on how to print the page.What is the structure of PCl5?
2) Structure of PCl5: It has a trigonal bipyramidal structure, in gaseous and liquid phases. The three equatorial P–Cl bonds are equivalent, while the two axial bonds are longer than equatorial bonds.How PCl5 is formed?
Phosphorus Penta chloride is prepared by passing an excess of dry chlorine into liquid trichloride. Chlorine reacts with phosphorus trichloride and solid phosphorus penta chloride is formed.What is phosphorus pentachloride used for?
PCl5 or Cl5P is used as an starting material for the synthesis of a variety of inorganic and organic phosphorus compounds used as pesticides, water treatment chemicals, flame retardants, phosphite antioxidants, plasticizers and stabilizers for plastic and elastomers, lube oil and paint additives.Is cucl2 ionic or covalent?
The bond that holds copper chloride together would be an ionic bond. Copper is a metal and chloride is a halogen. Usually, halogens like chlorine will react with metals to form a metal halide, an ionic compound.Why is PCl5 more reactive than PCl3?
PCl5 will be more reactive than PCl3 because the oxidation state of phosphorus in PCl5 is +5 whreas in PCl3 is +3. Therefore the replulsive force between the bonds is greater in PCl5 than in PCl3 and hence is more reactive.Why PCl3 is more stable than PCl5?
PCl3 is more stable than PCl5 because PCl5 has steric crowding(triagonal bipyramidal shape and sp3d2 hybridisation) than PCl3(triagonal planar structure and sp3 hybridisation).