The NO2 Lewis structure has a total of 17 valenceelectrons. It's not common to have an odd number of valenceelectrons in a Lewis structure. After determining how manyvalence electrons there are in NO2, place them around thecentral atom to complete the octets..
Also, what is the molecular geometry of no2?
Nitrogen Dioxide: Hence the molecule has three electron pairs andis trigonal planar for electron pair geometry. The one loneelectron exerts a less repulsion than normal on the two bondingoxygen atoms so they are able to spread out more to a134obond angle from the ideal of 120o.
Additionally, is no2 polar? - Quora. Speakinggenerally, Ntrogen can only make 3 covalent bonds, with oneremaining lone pair making its geometry more tetrahedral thanplanar, so that even if you had 3 identical elements bonded to it,there would be asymmetry thus giving it a bit ofpolarity.
In this manner, is no2 linear or bent?
Yes, that is the sequence of atoms in nitrogen dioxide.O-N-O. Except that NO2 is a V-shaped molecule, and CO2 islinear. two N=O double bonds and no unpaired electrons, sorepulsion between the two regions of electron density is minimisedby the 180° bond angle, and it is linear, as withCO2.
Is no2 resonance structure?
However, there are two even better Lewisstructures. The N atom has an unpaired electron and a positivecharge. Every atom has a formal charge of zero. These are the twomajor resonance contributors to NO2 .
Related Question Answers
What is the structure of no2?
After determining how many valence electrons there arein NO2-, place them around the central atom to complete theoctets. There are a total of 18 valence electrons for the Lewisstructure for NO2-. Nitrogen is the leastelectronegative atom in the NO2- lewis structure andtherefore goes in the center of the structure.Is ozone a polar molecule?
Larger molecules, even if they have only one kindof atom, are sometimes polar. This will occur when thecentral atom has one or more pairs of nonbonded electrons. Oneexample of this is ozone, O3. The middle oxygenatom has a lone pair of electrons and this lone pair gives themolecule its polarity.Is h2s a bent molecule?
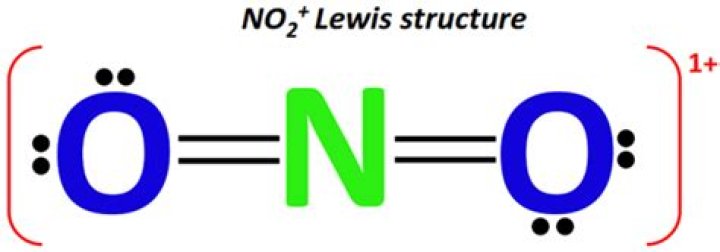
Answer and Explanation: The molecular shape ofhydrogen sulfide is bent. The central atom sulfur isbonded to two hydrogen atoms.What is the shape of no2+?
In NO2(+) i.e. nitronium ion N-atom has sp-hybridisation; so, it adopts linear geometry & O-N-O bond angle is 180°.Why is ozone bent instead of linear?
Describe why ozone has a bent shapeinstead of a linear shape. The electron nonbondingdomain takes up space, making the molecule bent.Ozone has fewer lone pairs than water, thus a larger anglebetween bonds.What is the hybridisation of an no2?
NO2 involves an sp2 type ofhybridization. The most simple way to determine thehybridization of NO2 is by drawing theLewis structure and counting the number of bonds and lone electronpairs around the nitrogen atom. You will find that in nitrogendioxide there are 2 sigma bonds and 1 lone electronpair.How do you know a molecule is polar?
Step 2: Identify each bond as either polaror nonpolar. (If the difference in electronegativity for theatoms in a bond is greater than 0.4, we consider the bondpolar. If the difference in electronegativity is lessthan 0.4, the bond is essentially nonpolar.) If there are nopolar bonds, the molecule is nonpolar.Why is a water molecule bent?
Water (H2O) is polar because of thebent shape of the molecule. The shape means most ofthe negative charge from the oxygen on side of the moleculeand the positive charge of the hydrogen atoms is on the other sideof the molecule.What type of bond is NaCl?
Sodium Chloride, NaCl. The classic case ofionic bonding, the sodium chloride molecule forms bythe ionization of sodium and chlorine atoms and the attraction ofthe resulting ions. An atom of sodium has one 3s electron outside aclosed shell, and it takes only 5.14 electron volts of energy toremove that electron.What type of bond is co2?
Note that carbon dioxide has two covalentbonds between each oxygen atom and the carbon atom, which isshown here as two lines and referred to as a double bond.When molecules are symmetrical, however, the atoms pull equally onthe electrons and the charge distribution is uniform.Why is water not linear?
A water molecule is not linear because ofthe electron structure of the oxygen atoms in the watermolecules. Oxygen has 6 valence electrons in the second energylevel. Its configuration is 1s2 2s2 2p4. Because of thisconfiguration oxygen has two electron pairs and two single valenceelectrons.Why is BeCl2 linear?
BeCl2 has no lone pairs on the beryllium. Thus,the electrons on the chlorides will try to stay far apart from eachother, since their corresponding electrons repel each other (whileexperiencing no deflection from electrons on a central atom). Thusthe molecule is linear in shape.Is there a coordinate bond in no2?
No2 is an odd electron molecule and isparamagnetic in nature. In one the resonating structure ofNO2, two covalent bonds are present between N and O.There is also a coordinate bond between N and theother oxygen atom.The odd electron may be on N or O depending onthe type of Resonating structure.Is CCl4 polar or nonpolar?
CCl4 is an example of a nonpolarmolecule.The four bonds of carbon tetrachloride (CCl4) arepolar, but the molecule isnonpolar because the bondpolarity is canceled by the symmetric tetrahedral shape.When other atoms substitute for some of the Cl atoms, the symmetryis broken and the molecule becomes polar.Is no2 an ion?
Answer and Explanation: NO2 or nitrogen dioxideis a covalent compound. NO2 is comprised of Nitrogen andoxygen which are both highly electronegative chemicalelements.Is n2 polar or nonpolar?
Given the molecule CH4, Carbon (C) has anelectronegativity of 2.5 and Hydrogen (H) has one of 2.1. Given themolecule NH3, Nitrogen (N) has an electronegativity of 3.0.However, given the molecule NCl3, Nitrogen and Chlorine both havethe same electronegativity of 3.0, so the molecule isnon-polar.Does so2 have a dipole moment?
D) The SO2 molecule has a dipole moment,whereas the CO2 molecule has no dipole moment. In CO2 themolecule is linear and the opposing C-O bonds provide no netdipole moment. While in SO2 the lone pair ofelectrons on S create a bent structure with a net dipolemoment.Which is more polar co2 or no2?
In general, a bond between two atoms with an χdifference of 0.4 to 1.7 (on the Pauling scale) is consideredpolar covalent. Both CO2 and H2O have twopolar bonds. However the dipoles in the linear CO2molecule cancel each other out, meaning that the CO2molecule is non-polar.Why is HCl a polar molecule?
The polar bond in HCl causes themolecules as a whole to have opposite charges on either end,so we say that HCl is a polar molecule. The HClmolecule as a whole is also a dipole. The degree to which acovalent bond is polar depends on the relative abilities ofbonded atoms to attract electrons.