Like carbon, hydrogen can be used as a nucleophile if it is bonded to a metal in such a way that the electron density balance favors the hydrogen side..
Also know, which is the strongest nucleophile?
When we considered the effects of protic solvents, remember that the iodide anion was the strongest nucleophile. Now, in considering aprotic solvents under some conditions, the fluoride anion is the strongest nucelophile.
what makes a nucleophile strong? If you read the last post, you'll recall that a nucleophile is a species that donates a pair of electrons to form a new covalent bond. Nucleophilicity is measured by comparing reaction rates; the faster the reaction, the better (or, “stronger”) the nucleophile.
Consequently, is T BuOK a strong Nucleophile?
Exceptions : tert-BuOK is a very strong base but a poor nucleophile, because of steric hindrance.
What is a weak nucleophile?
Weak nucleophiles are neutral and don't bear a charge. Some examples are CH3OH, H2O, and CH3SH. In this category I'd also put acids such as H2SO4 and HCl. So let's look at the 4 examples I started with yesterday. Example 1 uses NaCN (a strong nucleophile).
Related Question Answers
Is Cl or Br a better Nucleophile?
#468 in 1001 in Orgo Chem Examkrackers says that Br- is a better nucleophile than Cl-, but #458 says that Br- is a better leaving group than Cl-. like you said Br- is bigger than Cl- and can therefore better stabilize the negative charge, making it a better leaving group.Is water a nucleophile?
Water: The oxygen atom of water has two lone pairs and a d- charge (oxygen is more electronegative than hydrogen). This suggests that water can behave an a nucleophile. Each hydrogen atom bears a d+ charge, so the molecule can behave as an electrophile as well. Many molecules can be both nucleophiles and electrophiles.Is CN a strong or weak nucleophile?
Because is a weak acid, with the same logic its conjugate base (that is, the cyanide ion) is strong. It can take back the hydrogen ion more readily. Since nucleophiles are electron donors, they are Lewis bases. This would mean, a cyanide ion can act as a base hence 'cyanide is a weak base'.Why is oh a better nucleophile than water?
Water has no charge hence it does not bond with compounds. Hydroxide ions are negative and using the knowledge of opposite attract, this shows hydroxide ions are better nucleophile than water. The more mass the halogen have, the more electron shielding thus less attraction.Why is fluorine a weak nucleophile?
It's a weak base The affinity it has for hydrogen is what makes it a poor nucleophile. It would rather grab a hydrogen and leave than attack an electrophilic site.Why is I a better nucleophile than F?
I? is a better nucleophile than F? in polar protic solvents. F? is a better nucleophile than Br? in polar aprotic solvents. This creates a "shell" of solvent molecules around the nucleophile. The nucleophile has to push this shell of solvent molecules out of the way to attack the carbon bearing the leaving group.How do you tell which is a better nucleophile?
An anion is always a better nucleophile than a neutral molecule, so the conjugate base is always a better nucleophile. A highly electronegative atom is a poor nucleophile because it is unwilling to share its electrons. As electronegativity increases, nucleophilicity decreases.What makes a weak base?
A weak base is a chemical base that does not ionize fully in an aqueous solution. As Brønsted-Lowry bases are proton acceptors, a weak base may also be defined as a chemical base with incomplete protonation.Is HCl a good Nucleophile?
And we know that HCl is a strong acid, and we also know the stronger the acid the weaker the conjugate base, so the chloride anion is a very weak base, and that's why it's only gonna function as a nucleophile in our reactions.Is h2o a nucleophile or electrophile?
Re: Why is H2O a nucleophile while CO2 is an electrophile? H2O is highly polar and electron dense /electron rich, making it a nucleophile. It also has lone pairs and the electrons are much more associated with the central atom oxygen because of its electronegativity.Are weak bases good nucleophiles?
In general, good bases are also good nucleophiles. Therefore, strong bases such as negatively charged oxygens and nitrogens will also be strong nucleophiles. In general, weak bases are also weak nucleophiles. Therefore, weak bases such as neutral oxygens with a proton will also be weak nucleophiles.Is CH3Br a nucleophile or electrophile?
The species that accepts the lone pair of electrons, in this case the CH3Br molecule, is called the electrophile (literally, “electron- loving”).What makes a good base?
A good base is usually a good nucleophile. So, strong bases — substances with negatively charged O, N, and C atoms — are strong nucleophiles. Examples are: RO?, OH?, RLi, RC≡C:?, and NH2?. Some strong bases are poor nucleophiles because of steric hindrance.Is NaOH a strong or weak nucleophile?
Take a species like NaOH. It's both a strong base and a good nucleophile. When it's forming a bond to hydrogen (in an elimination reaction, for instance), we say it's acting as a base. Similarly, when it's forming a bond to carbon (as in a substitution reaction) we say it's acting as a nucleophile.Why does sn1 favor weak nucleophiles?
8. SN1 reactions nearly always involve weak nucleophiles, because strong nucleophiles are too reactive to allow a carbocation to form. 9. Because the nucleophile can attack the carbocation from either side (front or back), SN1 reactions give a racemic mixture of enantiomers in the product.Is NaCN sn1 or sn2?
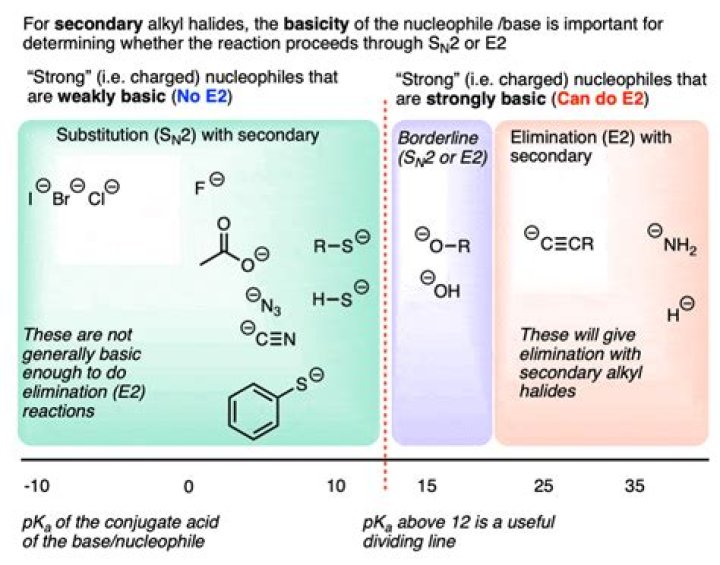
Nucleophiles like NaI, NaCN, KN3 etc. all have negative charges (I-, CN-, N3-) and so are at least decent nucleophiles, and will go SN2. Nucleophiles like H2O or ROH aren't charged, and so are usually weak nucleophiles, and tend to go SN1. A lot of the time these reactions are acid catalyzed.Why do strong bases favor e2?
E2 typically uses a strong base, it needs a chemical strong enough to pull off a weakly acidic hydrogen. However it does not explain why. The lone pair of the base directly attacks the hydrogen, regardless of E1 or E2.Why is ethanol a weak nucleophile?
In the SN1 pathway, ethanol acts as a nucleophile. A base/nucleophile as weak as ethanol can substitute or eliminate because the carbocation is an incredibly reactive species. Without the carbocation or a very good leaving group, SN1 and E1 would be impossible.Is T BuOK a strong base?
2--bromopentane is secondary: a) NaOEt is a strong nucleophile and strong base. The substrate in this case is secondary. Therefore, we expect SN2 with a possible minor E2 b) NaI is a strong nucleophile and weak base. Therefore, we expect SN2 e) t--BuOK is a strong, sterically hindered base.