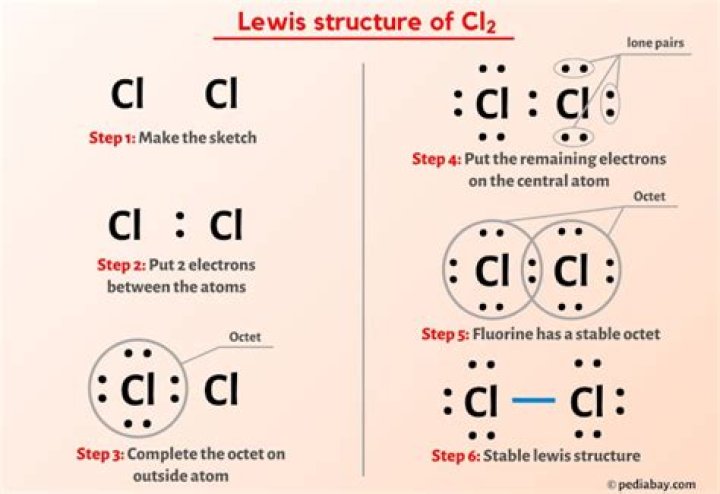
Cl2 is a diatomic molecule, whereas 2Cl means 2units of a negatively charged chlorine anion in a chemicalequation. The 2 in front of 2Cl just means that there are 2 singlechlorine ions. The 2 written in Cl2 means that thereare two chlorine atoms covalently bonded to form achlorine molecule..
Besides, what charge is cl2?
The resultant chlorine molecule or is neutral incharge as the two form covalent bonding. Thus they have nocharge as either of the two atoms has not lost or gained anyelectrons. Instead the one outer electron of each of the Cl atom isshared to form a covalent bond and a stable and neutral Chlorinemolecule .
Secondly, is cl2 a polar or nonpolar molecule? Why is an HCl molecule polar while a Cl2molecule is nonpolar? H & Cl have differentelectronegativities; therefore they form a polar bond. Cl& Cl have same electronegativity; therefore they form anonpolar bond. 3.
In this way, is cl2 an atom or molecule?
It is both. It is two atoms of the elementchlorine, chemically bonded together into a single chlorinemolecule. The term “element” is not the same asatom. An atom is the smallest unit of matter that hasa specific number of protons, neutrons, and electrons.
Is chlorine ion acidic?
In nature, chlorine is most abundant as achloride ion. Physiologically, it exists as an ion inthe body. The chloride ion is an essential anion that thebody needs for many critical functions. It also helps keep thebody's acid-base balance.
Related Question Answers
Is cl2 positive or negative?
If you add an electron, as is the case withchlorine, you upset the balance between the positiveand negative charges. There are 18 electrons and 17 protons,so the chlorine atom has become a charged chlorineion with a charge of negative one (-1).What is Sodiums charge?
Sodium atoms have no charge, sodiumhas eleven protons in its nucleus and eleven electrons orbiting it.This makes it electrically neutral. When sodium forms anion, it will form Na+ a cation. This is because sodium hasone valence electron (one electron in its outershell).What does 2cl mean?
Cl2 is a diatomic molecule, whereas 2Cl means 2units of a negatively charged chlorine anion in a chemicalequation. The 2 in front of 2Cl just means that thereare 2 single chlorine ions. These ions are NOT bonded to eachother.What is the name of cl2?
Chlorine is called Cl2 because it is adiatomic molecule. Diatomic means there are two atoms of the sameelements in the molecule. There are other diatomic molecule, suchas hydrogen (H2), nitrogen (N2), oxygen (O2), fluorine (F2),chlorine (Cl2), bromine (Br2), iodine(I2).How do you find the charge of an ion?
Subtract the number of electrons from the number ofprotons in an atom as a basic way of calculating the chargeof the ion. For example, if a sodium atom loses oneelectron, work out 11 - 10 = 1. A sodium ion has a +1charge, notated as Na+.What is the charge on a calcium ion?
Calcium also has a positive charge of 2.This makes it an ion. An ion is an atom of a chemicalelement that has an unequal number of electrons compared toprotons. Remember that electrons are negative charges, andprotons are positive charges.What is the oxidation state of cl2?
A/s Chemistry - Oxidation number & redox
| A | B |
| chlorine in Cl2 - oxidation number is | 0 |
| Cl in NaCl oxidation number is | -1 |
| oxidation number of O in its compounds (not with F)isalways | -2 |
| oxidation numbe rof F in its compounds is always | -1 |
What is the charge of aluminum?
Aluminum Ion Charge And Formula. Thecharge of an aluminum ion is typically 3+. This isbecause the element's atomic number is 13, reflecting the fact thatit has 13 electrons and 13 protons.What type of bond is HCl?
HCl ( Hydrogen Chloride ) is a binarycompound consisting of two atoms, a hydrogen ( H ) and aChlorine (Cl ) atom. Both of these are non-metallic atoms and sowill form a covalent bond between them.Is Co a molecule?
Molecules containing more than two atoms aretermed polyatomic molecules, e.g., carbon dioxide(CO2) and water (H2O). Polymermolecules may contain many thousands of component atoms. Awater molecule is made up of two hydrogen atoms and oneoxygen atom.What is a group of molecules called?
A molecule is two or more atoms chemically joinedtogether. If at least two different elements make up themolecule, it is called a compound. For example,hydrogen gas (H2) and water (H2O) are molecules, but wateris also a compound because it is made up of hydrogen andoxygen.Is so2 diatomic?
2 Answers. One atom of oxygen is a fictional speciesoutside of a plasma. Oxygen is naturally diatomic, and theprocess by which it oxidizes sulfur is radical-based, leading toboth oxygen atoms reacting. The fact that SO2 forms insteadof SO is a result of the [redundant answer] thermodynamics favorSO2 over SO.Is cl2 an element?
Chlorine by itself is an element. Thisis chlorine element with one extra electron. Chlorinegas, Cl2, is a molecule. The most common compound ofchlorine is sodium chloride, NaCl (table salt.)Why is oxygen a diatomic molecule?
Oxygen generally exists as a diatomicmolecule in the atmosphere when it is not combined with anyother element. It forms the molecule O2 because in thatconfiguration, it has its lowest energy level when uncombined. Inthe standard model of the oxygen molecule, eachoxygen atom shares two pairs of electrons.Is o2 an atom or molecule?
Oxygen in the atmosphere is a moleculebecause it contains molecular bonds. It is not a compoundbecause it is made from atoms of only one element -oxygen. This type of molecule is called a diatomicmolecule, a molecule made from two atoms ofthe same type.Is chlorine a diatomic molecule?
Diatomic elements are pure elements that formmolecules consisting of two atoms bonded together. There areseven diatomic elements: hydrogen, nitrogen, oxygen,fluorine, chlorine, iodine, bromine. For example, oxygen canexist as the triatomic molecule, ozone.Is gold an atom or molecule?
Gold, for example, is made of only goldatoms. When matter is made of only one kind of atom, itis called an element.Why is HCl polar and cl2 nonpolar?
Why is an HCl molecule polar while aCl2 molecule is nonpolar? H & Cl have differentelectronegativities; therefore they form a polar bond.Electrons are more attracted to one atom in a polarmolecule; however they do not completely transfer from one atom toanother.Is ClF dipole dipole?
The ClF bond is a polar covalent bond and themolecule has a net dipole moment (unlike CCl4 forinstance where the bond dipoles cancel giving the moleculeno net dipole moment). The intermolecular interactions willbe made up of dipole-dipole interactions anddispersion interactions.