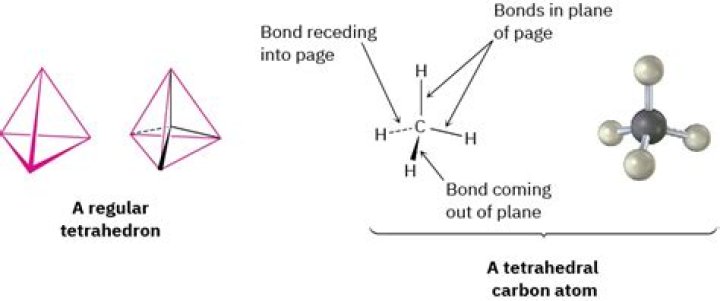
Carbon: always tetravalent and often tetrahedral Atoms combine in many different ways; we have already seen an example of how a covalent bond can form between two hydrogen atoms, producing molecular (H2), as opposed to the atomic form of hydrogen..
Keeping this in view, why does carbon have a tetrahedral geometry?
The carbon has 4 valence electrons and thus needs 4 more electrons from four hydrogen atoms to complete its octet. The hydrogen atoms are as far apart as possible at 109o bond angle. This is tetrahedral geometry. This molecule provides the basis for the tetrahedral geometries at each carbon in a hydrocarbon chain.
what molecule has a tetrahedral shape? Tetrahedral Geometry Molecules of methane, CH4, ammonia, NH3, and water, H2O, all have four electron groups around their central atom, so they all have a tetrahedral shape and bond angles of about 109.5°.
Keeping this in consideration, what is a tetrahedral carbon?
Illustrated Glossary of Organic Chemistry - Tetrahedral carbon. Tetrahedral carbon: A carbon atom with four attachments, and bond angles of approximately 109.5o. The overall shape is that of a tetrahedron (i.e., a pyramid with all faces being equilateral triangles, or nearly so).
Why do alkanes have a tetrahedral shape?
The tetrahedral geometry is created because every single sp3 electron from the carbon repels the other with an equal coulombic force (wouldn't be equal if the carbon wasn't hybridized) in the s¹p³ (sp³) orbital. This creates an equal distribution to the atoms and gives it a tetrahedral geometry.
Related Question Answers
Why does carbon bond so easily?
Because each carbon is identical, they all have four valence electrons, so they can easily bond with other carbon atoms to form long chains or rings. In fact, a carbon atom can bond with another carbon atom two or three times to make double and triple covalent bonds between two carbon atoms.How many angles are there in the tetrahedral structure?
109.5 degrees
Is h2o an organic molecule?
Molecules associated with living organisms are organic. These include nucleic acids, fats, sugars, proteins, enzymes and hydrocarbon fuels. Inorganics include salts, metals, substances made from single elements and any other compounds that don't contain carbon bonded to hydrogen. So, WATER is basically inorganic.Is h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.Should all angles in methane be equal?
Methane is a tetrahedral shape molecule. All the angles formed by the centre of the tetrahedral are equal. Thus all CH lengths and HCH angles in methane are equal. If you have any doubts, you could ask it in comments.What is the bond angle of h2o?
104.5o
Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Is Diamond a tetrahedral?
In a diamond, the carbon atoms are arranged tetrahedrally. Each carbon atom is attached to four other carbon atoms 1.544 x 10-10 meter away with a C-C-C bond angle of 109.5 degrees. Because of its tetrahedral structure, diamond also shows a great resistance to compression.Does water have a tetrahedral structure?
Water structure However, in liquid water or in ice, the lone pairs form hydrogen bonds with neighboring water molecules. The most common arrangement of hydrogen atoms around an oxygen is tetrahedral with two hydrogen atoms covalently bonded to oxygen and two attached by hydrogen bonds.Is a tetrahedral symmetrical?
The symmetric shapes are linear, trigonal planar, and tetrahedral. The unsymmetric shapes are bent and trigonal pyramidal. The symmetric shapes are associated with molecules whose central atoms have no lone pairs. The unsymmetric shapes are associated with molecules whose central atoms have one or more lone pairs.Is ammonia a tetrahedral?
Ammonia has 4 regions of electron density around the central nitrogen atom (3 bonds and one lone pair). These are arranged in a tetrahedral shape. The resulting molecular shape is trigonal pyramidal with H-N-H angles of 106.7°.How do you determine polarity?
Step 2: Identify each bond as either polar or nonpolar. (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.Is XeF4 tetrahedral?
But here Xe atom have two lone pair even after bonding with 4 fluorine ions. So the hybridization will be sp3d2. Also due to the presence of lone pair would be distorted due to the electron dense cloud of the lone pairs. So the shape of XeF4 would be square planar rather than tetrahedral.What comes after tetrahedral numbers?
The n -th tetrahedral number is the sum of the first n triangular numbers added up. The first few tetrahedral numbers (sequence A000292 in OEIS) are: 1, 4, 10, 20, 35, 56, 84, 120, 165, 220, 286, 364, 455, 560, 680, 816, 969, …What is planar structure?
Illustrated Glossary of Organic Chemistry - Planar. Planar: Said of a molecule when all of its atoms lie in the same plane. Can also be said for a portion of a molecule, such as a ring. Atoms, groups, bonds, or other objects lying within the same plane are periplanar or coplanar. Lewis structure.Is water tetrahedral or bent?
An example of bent molecular geometry that results from tetrahedral electron pair geometry is H2O. The water molecule is so common that it is wise to just memorize that water is a BENT molecule. The oxygen has 6 valence electrons and thus needs 2 more electrons from 2 hydrogen atoms to complete its octet.Why is it called octahedral?
In chemistry, octahedral molecular geometry describes the shape of compounds with six atoms or groups of atoms or ligands symmetrically arranged around a central atom, defining the vertices of an octahedron. The octahedron has eight faces, hence the prefix octa.How do you know if something is tetrahedral?
The total number of bonds and lone electron pairs determine the steric number of the molecule. Tetrahedral molecules have a steric number of four because they have four bonds and no lone electron pairs. The presence of lone electron pairs affects the shape of the molecule.How many tetrahedral angles does a molecule of methane has?
six tetrahedral angles